WSRC-TR-2000-00353
Characterization of Tank 40H Supernate and
Hydroxide
Washing of Sludge
W. R. Wilmarth, V. H. Dukes, J. T. Mills, C. J. Coleman, J.
C. Hart,
W. T. Boyce, C. C. Diprete, and R. L. Cadle
Westinghouse Savannah River Company
Aiken, SC 29808
This report was prepared as an account of work sponsored by an agency of the United States Government. Neither the United States Government nor any agency thereof, nor any of their employees, makes any warranty, express or implied, or assumes any legal liability or responsibility for the accuracy, completeness, or usefulness of any information, apparatus, product or process disclosed, or represents that its use would not infringe privately owned rights. Reference herein to any specific commercial product, process or service by trade name, trademark, manufacturer, or otherwise does not necessarily constitute or imply its endorsement, recommendation, or favoring by the United States Government or any agency thereof. The views and opinions of authors expressed herein do not necessarily state or reflect those of the United States Government or any agency thereof.
This report has been reproduced directly from the best available copy.
Available for sale to the public, in paper, from: U.S. Department of Commerce, National Technical Information Service, 5285 Port Royal Road, Springfield, VA 22161, phone: (800) 553-6847, fax: (703) 605-6900, email: orders@ntis.fedworld.gov online ordering: http://www.ntis.gov/support/ordering.htm
Available electronically at http://www.osti.gov/bridge/
Available for a processing fee to U.S. Department of Energy and its contractors, in paper, from: U.S. Department of Energy, Office of Scientific and Technical Information, P.O. Box 62, Oak Ridge, TN 37831-0062, phone: (865 ) 576-8401, fax: (865) 576-5728, email: reports@adonis.osti.gov
Summary
During June 2000, the 3H Evaporator system is scheduled to receive wash water from washing the sludge and supernate currently in Tank 40H. The supernate from Tank 40H contains concentrated supernate from Tank 38H, the 2H Evaporator drop tank. This material may contain soluble silicon from the DWPF recycle stream. Therefore, SRTC examined the contents of Tank 40H and simulated the hydroxide wash of the sludge. The results of these tests indicate the following conclusions.
Introduction
The High Level Waste Tank Farms store and process high-level liquid wastes from a number of sources including F- and H-Canyons and a recycle stream from the Defense Waste Processing Facility (DWPF). However, during processing of Defense Waste Processing Facility Recycle stream, difficulty in flow in the Gravity Drain Line (GDL) occurred in July 1997. Video inspection indicated solid deposits present in the Gravity Drain Line. Laboratory experiments showed that compounds of the general formula, Na8Al6Si6O24(NO3)2· 4H2O, form readily under the evaporator conditions.,, During November 1999 a sample of solids removed from the Evaporator cone contained mostly sodium aluminosilicate.
Due to the abundance of aluminum in the high-level liquid wastes, silicon is the limiting reagent for formation of the aluminosilicate solid phase. The primary source for silicon is from frit return to the tank farm in the DWPF recycle stream. The deposition of sodium aluminosilicate along with sodium diuranate in the 242-16H evaporator system led to the removal of authorization to process High Level Waste containing DWPF recycle. Therefore, High Level Waste Engineering requested SRTC to perform analysis of the supernate contents of Tank 40H and pre and post samples from sludge washing to ensure silicon levels are sufficiently low to allow processing of the supernate through the 3H Evaporator.
Additionally, chemical and radiochemical characterization of the Tank 40H sludge and supernate was performed to support the planned washing. In particular, the chemical constituency of the sludge was explored prior to washing and after the washing treatment. The free hydroxide concentration of the Tank 40H supernate was also measured.
Experimental
The SRTC received two surface samples from Tank 40H numbered ESP-238 and ESP-239. Additionally, SRTC received a sample containing sludge (ESP-237). The samples from the tanks entered B-block of the Shielded Cells upon receipt. Personnel determined the density of the liquid samples by emptying the contents of the dip bottles into pre-weighed 100-mL graduated cylinders with the accuracy of the graduated cylinders verified to ± 0.5% at 100 mL prior to use. Personnel removed representative aliquots from the surface samples, filtered through a 0.45 micron filter, and diluted into 1 M nitric acid by a factor of ~50. For the free hydroxide measurement, the filtered supernate was diluted 10-fold into distilled water and submitted for analysis.
A large aliquot of the sludge sample was dried at 100 ° C to determine the weight % solids from a given volume of sludge. Three samplings of the dried solids were submitted for aqua regia dissolution and three others were submitted for cesium hydroxide fusion dissolution with full characterization referenced below.
The sludge washing experiment was conducted as follows. A 10-mL aliquot of the well-mixed sludge sample (ESP-237) was transferred to a 40-mL graduated centrifuge tube. Twenty milliliters of caustic water (nominally 1 M sodium hydroxide and 0.4 M sodium nitrate) was added to the centrifuge tube. This composition simulates the predicted final composition of the wash water at the end of the first wash. Each tube was shaken vigorously for 10 minutes to thoroughly mix the sludge and wash water. Afterward, the tubes were placed into the centrifuge for 10 minutes at 2000 rpm. Twenty-five milliliters of clear liquid was removed from the tube with portions removed from the cell without dilution for analysis. The remaining washed sludge was dried, sampled for each dissolution method and submitted for analysis.
Many of the confirmation samples were put through a filtering protocol to determine the total amount of silicon in a given volume of liquid waste. The protocol included sampling as-received waste, sequential filtration through at least 0.2 micron and 0.02 micron filters, and analyzing filtrates and filters for aluminum and silicon. In some instances 0.45 micron filters were, also, used. Tables of data are presented with the following information: Si concentration (mg/L) in diluted sample, corrected Si concentration (mg/L, Si diluted x dilution factor), Si concentration (mg/L) from digested filter, Si amount (mg) on filter (Si concentration * 0.05 L volume) and Si concentration total (mg/L, [Si]0.02 filtrate + S Sifilter/Sample volume).
The Analytical Development Section performed the following analyses: Inductively Coupled Plasma – Mass Spectrometry, Inductively Coupled Plasma – Emission Spectroscopy, Gamma Spectroscopy, and Alpha Pulse Height Analysis.
Results and Discussion
Analysis of Tank 40H Supernate
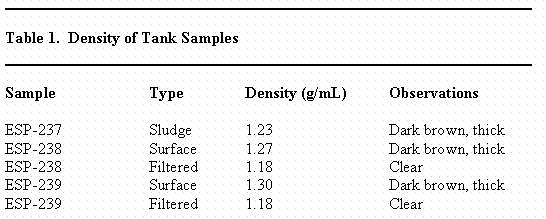
Table 1 contains the measured density and a brief description of the samples as received from Tank 40H. For Tank 40H, SRTC received two surface samples (ESP-238 and ESP-239). The density measured 1.27 and 1.30 g/mL for the surface samples (ESP-238 and ESP-239, respectively) prior to filtration and 1.18 g/mL after filtering through a 0.45 micron filter.

Tables 2 and 3 (located in Appendix 1) contain the elemental analysis of the liquid samples from Tank 40H. The authors corrected the concentrations reported for the original 50X dilution. Sodium ion concentration in Tank 40H measured 4.4 and 4.5 M for the surface samples. Silicon concentrations measured very low at 21.2 and 23.4 mg/L for the samples ESP-238 and ESP-239, respectively. Aluminum concentrations were much higher and measured 5.32 x 103 and 5.71 x 103 mg/L, respectively. Therefore, the calculated Ksp for the formation of aluminosilicate in these solution phases is 1.5 x 10-4 M2 for sample ESP-238 and 1.8 x 10-4 M 2 for sample ESP-239. These values of the Ksp clearly indicate that the solution is at equilibrium. Using the model developed for other tanks in H-Tank Farm, the predicted solids generation during a three-fold concentration in the 3H Evaporator would be 0.08 g/L. This solids formation (0.1 g/L) is very similar to the current 3H Evaporator feed. Additionally, this neglects a dilution by the large amount of inhibited water added to wash the sludge. Therefore, it appears from an aluminosilicate formation perspective that processing the existing supernate in Tank 40H would be no different than the current feed to the 3H. The next aspect examined was the silicon concentration in the sludge and in the wash water after sludge washing.
The uranium concentration was measured for diluted sub-samples from the filtered supernate samples (ESP-238 and ESP-239). The dilution-corrected concentrations are also shown in Table 2. The uranium concentration is very low and measured only 2 mg/L on average. The % enrichment is also very low and measured 1.3 based on analysis of the sub-sample from ESP-239. No 235U were detected in the 50:1 sample of ESP-238.
Characterization of Tank 40H Sludge
Table 1 contains the density information for the sludge sample removed from Tank 40H (ESP-237) prior to the addition of inhibited water to wash the sludge. The density of the sludge slurry was 1.23 g/L. An aliquot of the sludge slurry (~8 mL, 9.258 g) was dried in a Teflon beaker at 110 ° C oven overnight until a constant weight was obtained. The net dried weight of the sludge was 2.940 g yielding wt % dried solids of 31.8 %.

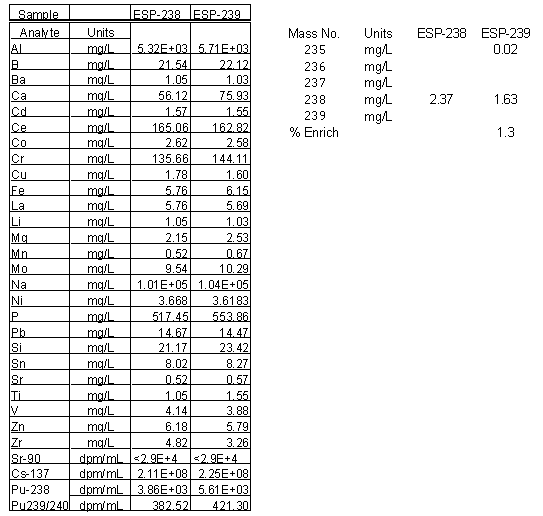
Table 4 contains the elemental results from the ICP-ES analysis of samples of the as-received sludge (labeled ESP-237-1 and ESP-237-2). Samples of dried as-received sludge were dissolved by two different methods (cesium hydroxide fusion and aqua regia dissolution). Silicon was present at less than 1 wt % with measured concentrations averaging 0.33 wt % for the aqua regia dissolution samples and 0.43 wt % for the cesium hydroxide fusion method. These silicon levels are similar to that measured by Hay where the silicon concentration in Tank 51H sludge measured 0.57 wt %. The aluminum and iron concentration in the as-received and dried Tank 40H sludge was also lower than that measured in Tank 51H sludge. The aluminum and iron concentrations in the Tank 40H dried sludge slurry measured approximately 3.35 and 10.6 wt %, respectively. The iron concentration is well less than the 22.1 wt % measured for the Tank 51 sludge.
Table 3 in the Appendix includes the measured actinide concentrations in the dried sludge slurry from Tank 40H. The uranium concentration measured ~1.7 wt %, again, slightly lower than the ~3 wt % in the Tank 51H sludge measured by Hay and Bibler. Likewise, the other radionuclide concentrations are also lower. For example, the 90Sr concentration average 375 m Ci/g in Tank 40H compared to 564 m Ci/g in the Tank 51H sludge.
Sludge Washing and Silicon Leach
To assess the silicon behavior during the washing step, two portions of the as-received sludge slurry (10 mL) were added to 20 mL of a non-radioactive, caustic simulant that was 1 M sodium hydroxide and 0.4 M sodium nitrate. The simulant composition was intended to simulate the final concentration of the supernate after the first washing step. No measures were taken to remove the supernate in the sludge slurry, therefore, the resultant wash water would be more aggressive (high hydroxide concentration) at dissolving silicon from the sludge. Therefore, this test would provide a bounding assessment at silicon removal from the sludge. Samples from the wash water were removed from the shielded cells without dilution to ensure a low detection limit for silicon.
Table 4, also, includes the elemental composition of the washed sludge as measured from cesium hydroxide digestion method. The results show agreement that silicon is not washed from the sludge. The silicon concentration rises from 0.3 – 0.4 wt % in the as-received dried sludge slurry to 1.1 wt % on average in the dried wash sludge. The concentration of silicon in the sludge should rise if silicon had not been washed from the sludge along with other soluble species. This confirmation of the analysis of the wash water and the original supernate in Tank 40H indicates those two results. The first result is that the existing supernate is at equilibrium with respect to aluminosilicate formation (as expected). The second indicates that washing the sludge with inhibited water will not add silicon to the system and disturb the equilibrium with respect to aluminosilicate formation.
Other observations concerning the data for the washed sludge from Table 3 (in Appendix) and Table 4 include the following. The sodium ion concentration in the sludge will not be significantly reduced during the first washing step. The sodium ion concentration in the Tank 40H sludge measured 22.25 wt % after washing compared to 21.5 wt % initially. The iron concentration, however, did increase as expected and rose from 10.6 wt % to 15.6 wt %. Likewise, the uranium concentration increased to 2.4 wt % from 1.7 wt % initially.
Confirmation Samples
The first set of confirmation samples (ESP-245 and ESP-246, taken after 7 days from pump operation At an elevation of 194") were received at SRTC and placed into the Shielded Cells. The sample ESP-245 was opened and duplicate dilutions were performed into dilute nitric acid. The results for these samples initially showed silicon concentrations 316 and 348 mg/L. Inspection of the acidic subsample showed evidence of particulate matter dispersed in the sample. Table 5 contains the results of additional analyses of the confirmation samples. Attempts were made to avoid getting solids into the subsamples during analyses. The silicon concentration in these samples averaged 127.2 mg/l.
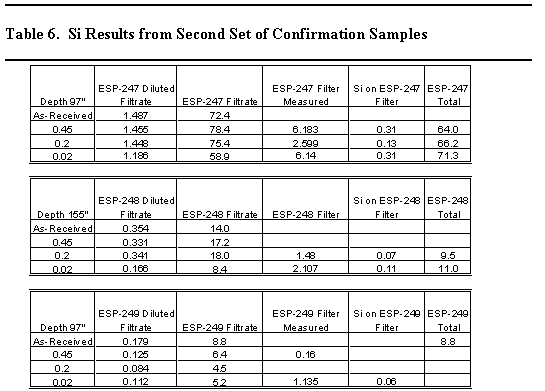
The silicon concentrations in the confirmation sample were not anticipated based on the work presented above. Therefore, a second series of samples were taken after ~ 25 days at two elevations in the tank. The samples were analyzed as received and after sequential filtrations. The filters were also digested using a sodium peroxide fusion.
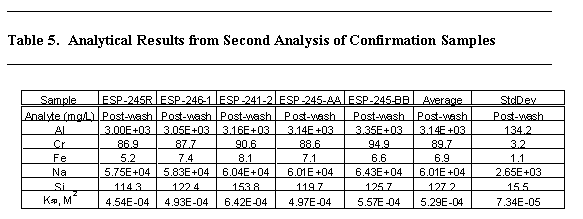
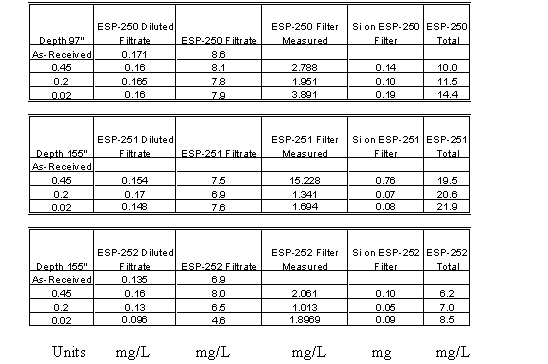
Table 6 contains the results of those analyses. Experimental difficulties were encountered with two of the samples (ESP-248 and ESP-249) and precluded complete analysis of these samples. However, the total silicon concentrations were quite low with the exception of one sample taken at an elevation of 97" from the bottom of the tank. The silicon concentration in that sample (ESP-247) measured 71 mg/L. Figure 1 shows a plot of the silicon concentration in Tank 40H with the time zero point being the end of the first washing cycle. The data strongly suggests that the silicon concentrations are arising from very small particulate silicon-containing materials.
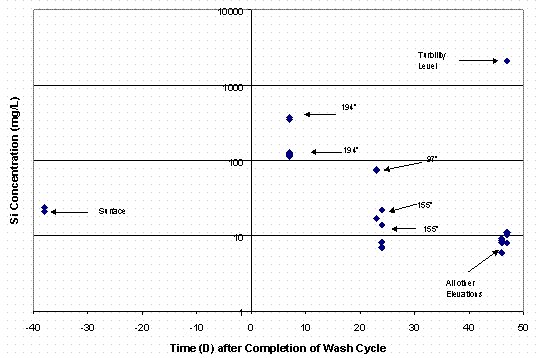
Figure 1. Silicon Concentrations in Tank 40H
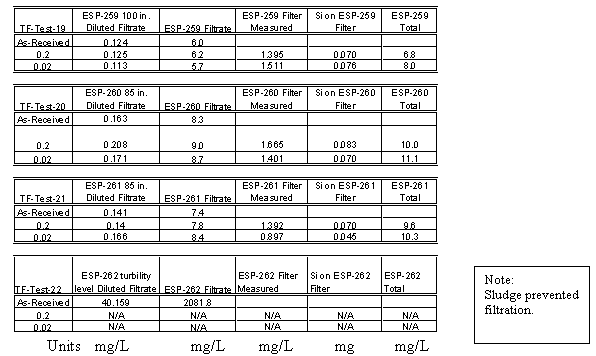
The results from the third set of samples indicated that the tank contents were settling and that given sufficient time the decant from Tank 40H could be performed that would ensure low silicon concentration. A third set of samples was pulled from Tank 40H in an effort to measure a silicon profile of the tank. Table 7 contains the results of these analyses. The data is also included in Figure 1. The results show that the silicon concentrations are very low, even down to an elevation of 85" off the bottom of the tank with a sludge layer at about 50 – 60". A sample taken from the sludge layer showed concentration on the order of 2 g/L silicon. This data confirms that with a transfer jet at 100 to 110" the liquid phase decanted from the tank would contain very low amounts of silicon, similar to concentrations in normal canyon waste. Figure 2 shows the data presented as a vertical profile in the tank.
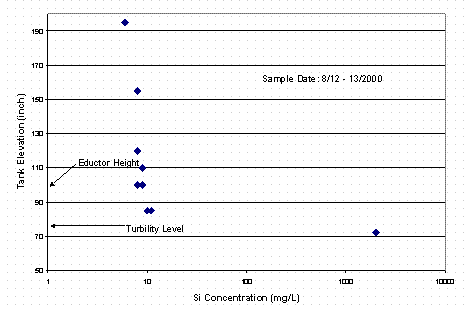
Figure 2. Si Concentration Profile in Tank 40H
Conclusions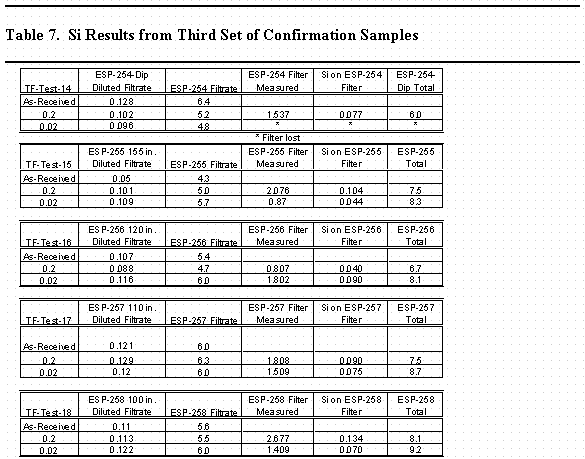
The 3H Evaporator system will receive influent from washing the sludge and supernate currently in Tank 40H. The supernate from Tank 40H contains concentrated supernate from Tank 38H, the 2H Evaporator drop tank. This material may contain soluble silicon from the DWPF recycle stream. Therefore, SRTC examined the contents of Tank 40H and simulated the hydroxide wash of the sludge.
Analysis of the supernate from Tank 40H prior to sludge washing showed very low silicon levels (~20 mg/L) and high soluble aluminum concentrations (0.2 M). At these levels, the supernate is in equilibrium with regards to sodium aluminosilicate formation. Processing this supernate from Tank 40H through the 3H Evaporator prior to the addition of large quantities of inhibited water would produce the same amount of aluminosilicate solids as the current Tank 32H feed.
Analysis of dried sludge slurry showed the silica concentration to be between 0.3 and 0.4 wt %. Samples of the dried sludge were washed with a salt solution and silicon was not detected in the wash water simulating the Tank 40H sludge washing step and indicates that silicon will not be leached from the sludge. This confirmation of the analysis of the wash water and the original supernate in Tank 40H indicates those two results. The first result is that the existing supernate is at equilibrium with respect to aluminosilicate formation (as expected). The second indicates that washing the sludge with inhibited water will not add silicon to the system and disturb the equilibrium with respect to aluminosilicate formation.
Confirmation samples taken after the washing of the sludge in Tank 40H indicate that fine particulates containing silicon are suspended during the washing step. Analysis of samples taken at different intervals following suspension of pump operation shows settling of these particles. In order to process the wash water from the sludge washing operation it is prudent to allow the tank to settle prior to decant to avoid entraining silicon species during transfer.
Acknowledgements
The authors wish to express appreciation to the following personnel for their contributions to this work. Personnel include L. Farrow (ADS), R. Peterson (calculations), N. Bibler, M. Hay, and T. Fellinger (discussions) and many others.
Appendix
Table 3. Mass Spectral Data from Tank 40 H Samples
|
Sample |
Wash Decant 1 |
Wash Decant 2 |
ESP-237-1 |
ESP-237-2 |
ESP-237-1 |
ESP-237-2 |
Washed Sludge 1 |
Washed Sludge 2 |
||||||||
|
CsOH |
CsOH |
Aq Regia |
Aq Regia |
CsOH |
CsOH |
|||||||||||
|
Mass |
Units |
Units |
||||||||||||||
|
82 |
microg/L |
18.64 |
19.85 |
Wt % |
|
|
|
|
|
|
||||||
|
84 |
microg/L |
Wt % |
|
|
|
|
2.84E-03 |
2.62E-03 |
||||||||
|
85 |
microg/L |
0.00 |
0.00 |
Wt % |
0.00E+00 |
0.00E+00 |
0.00E+00 |
0.00E+00 |
6.45E-04 |
6.76E-04 |
||||||
|
86 |
microg/L |
Wt % |
4.98E-04 |
6.27E-04 |
4.88E-04 |
6.42E-04 |
1.65E-03 |
1.51E-03 |
||||||||
|
87 |
microg/L |
228.35 |
243.13 |
Wt % |
1.53E-03 |
1.58E-03 |
1.50E-03 |
1.61E-03 |
6.13E-03 |
6.82E-03 |
||||||
|
88 |
microg/L |
1.42 |
1.87 |
Wt % |
5.03E-03 |
5.20E-03 |
4.93E-03 |
5.33E-03 |
9.10E-04 |
6.71E-04 |
||||||
|
89 |
microg/L |
Wt % |
7.63E-04 |
6.19E-04 |
7.48E-04 |
6.34E-04 |
6.06E-01 |
2.35E-01 |
||||||||
|
90 |
microg/L |
8.22 |
15.66 |
Wt % |
1.48E+00 |
2.62E-01 |
1.45E+00 |
2.68E-01 |
1.35E-01 |
5.52E-02 |
||||||
|
91 |
microg/L |
Wt % |
2.99E-01 |
5.57E-02 |
2.93E-01 |
5.71E-02 |
2.30E-01 |
6.97E-02 |
||||||||
|
92 |
microg/L |
89.92 |
97.00 |
Wt % |
7.04E-01 |
9.94E-02 |
6.90E-01 |
1.02E-01 |
1.30E-03 |
1.03E-03 |
||||||
|
93 |
microg/L |
2.01 |
9.16 |
Wt % |
1.41E-02 |
7.36E-04 |
1.39E-02 |
7.54E-04 |
2.29E-01 |
9.30E-02 |
||||||
|
94 |
microg/L |
52.17 |
57.95 |
Wt % |
6.64E-01 |
9.50E-02 |
6.50E-01 |
9.73E-02 |
1.23E-04 |
1.38E-04 |
||||||
|
95 |
microg/L |
543.43 |
564.82 |
Wt % |
7.56E-04 |
|
7.41E-04 |
|
3.62E-02 |
1.54E-02 |
||||||
|
96 |
microg/L |
97.80 |
106.79 |
Wt % |
9.80E-02 |
1.64E-02 |
9.60E-02 |
1.67E-02 |
|
|
||||||
|
97 |
microg/L |
470.11 |
490.44 |
Wt % |
5.34E-04 |
|
5.24E-04 |
|
1.31E-04 |
1.51E-04 |
||||||
|
98 |
microg/L |
563.82 |
585.40 |
Wt % |
7.83E-04 |
1.94E-04 |
7.67E-04 |
1.99E-04 |
9.11E-04 |
9.25E-04 |
||||||
|
99 |
microg/L |
584.61 |
616.12 |
Wt % |
8.02E-04 |
1.04E-03 |
7.86E-04 |
1.07E-03 |
|
1.56E-04 |
||||||
|
100 |
microg/L |
507.32 |
531.45 |
Wt % |
5.81E-04 |
|
5.69E-04 |
|
|
|
||||||
|
101 |
microg/L |
74.68 |
79.24 |
Wt % |
|
|
|
|
|
|
||||||
|
102 |
microg/L |
68.12 |
74.89 |
Wt % |
|
|
|
|
3.92E-04 |
4.20E-04 |
||||||
|
103 |
microg/L |
45.40 |
46.33 |
Wt % |
4.47E-04 |
5.68E-04 |
4.38E-04 |
5.81E-04 |
|
|
||||||
|
104 |
microg/L |
60.30 |
62.59 |
Wt % |
|
6.58E-05 |
|
6.74E-05 |
4.73E-04 |
4.87E-04 |
||||||
|
105 |
microg/L |
224.17 |
232.57 |
Wt % |
5.76E-04 |
6.19E-04 |
5.65E-04 |
6.34E-04 |
9.56E-03 |
5.54E-03 |
||||||
|
106 |
microg/L |
202.35 |
207.72 |
Wt % |
2.07E-02 |
4.58E-03 |
2.02E-02 |
4.69E-03 |
9.08E-03 |
7.42E-03 |
||||||
|
107 |
microg/L |
186.19 |
203.68 |
Wt % |
9.43E-03 |
5.10E-03 |
9.25E-03 |
5.23E-03 |
4.11E-03 |
2.83E-03 |
||||||
|
108 |
microg/L |
92.54 |
95.09 |
Wt % |
8.16E-03 |
2.09E-03 |
7.99E-03 |
2.14E-03 |
6.70E-03 |
5.96E-03 |
||||||
|
109 |
microg/L |
63.21 |
76.85 |
Wt % |
3.91E-03 |
3.31E-03 |
3.83E-03 |
3.39E-03 |
2.03E-02 |
1.93E-02 |
||||||
|
110 |
microg/L |
48.06 |
48.10 |
Wt % |
1.85E-02 |
1.36E-02 |
1.81E-02 |
1.39E-02 |
1.82E-02 |
1.89E-02 |
||||||
|
111 |
microg/L |
24.17 |
25.74 |
Wt % |
1.16E-02 |
1.29E-02 |
1.14E-02 |
1.32E-02 |
3.30E-02 |
3.48E-02 |
||||||
|
112 |
microg/L |
45.51 |
47.96 |
Wt % |
2.23E-02 |
2.42E-02 |
2.19E-02 |
2.48E-02 |
3.84E-02 |
4.08E-02 |
||||||
|
113 |
microg/L |
17.62 |
23.13 |
Wt % |
1.01E-02 |
1.19E-02 |
9.93E-03 |
1.22E-02 |
||||||||
|
114 |
microg/L |
46.14 |
47.42 |
Wt % |
2.13E-02 |
2.41E-02 |
2.08E-02 |
2.46E-02 |
9.65E-03 |
1.02E-02 |
||||||
|
115 |
microg/L |
Wt % |
|
|
|
|
|
|
||||||||
|
116 |
microg/L |
51.13 |
54.06 |
Wt % |
6.02E-03 |
7.01E-03 |
5.90E-03 |
7.18E-03 |
||||||||
|
117 |
microg/L |
32.42 |
35.88 |
Wt % |
|
|
|
|
3.05E-04 |
3.17E-04 |
||||||
|
118 |
microg/L |
98.02 |
101.00 |
Wt % |
|
|
|
|
3.29E-03 |
3.66E-03 |
||||||
|
119 |
microg/L |
52.00 |
53.31 |
Wt % |
3.20E-03 |
4.20E-03 |
3.13E-03 |
4.30E-03 |
3.73E-04 |
3.29E-04 |
||||||
|
120 |
microg/L |
126.73 |
136.10 |
Wt % |
|
|
|
|
4.19E-04 |
3.70E-04 |
||||||
|
121 |
microg/L |
15.10 |
11.62 |
Wt % |
|
|
|
|
|
|
||||||
|
122 |
microg/L |
23.32 |
22.66 |
Wt % |
|
|
|
|
3.07E-04 |
2.63E-04 |
||||||
|
123 |
microg/L |
15.35 |
8.59 |
Wt % |
|
|
|
|
1.42E-04 |
|
||||||
|
124 |
microg/L |
27.09 |
28.27 |
Wt % |
|
|
|
|
|
|
||||||
|
125 |
microg/L |
Wt % |
|
|
|
|
|
|
||||||||
|
126 |
microg/L |
9.18 |
8.76 |
Wt % |
|
|
|
|
|
|
||||||
|
127 |
microg/L |
143.97 |
170.65 |
Wt % |
|
|
|
|
|
|
||||||
|
128 |
microg/L |
Wt % |
|
|
|
|
|
|
||||||||
|
129 |
microg/L |
Wt % |
|
|
|
|
1.72E-04 |
1.70E-04 |
||||||||
|
130 |
microg/L |
0.32 |
Wt % |
1.74E-04 |
1.21E-04 |
1.71E-04 |
1.24E-04 |
|
|
|||||||
|
131 |
microg/L |
Wt % |
|
|
|
|
9.93E-04 |
9.25E-04 |
||||||||
|
132 |
microg/L |
Wt % |
4.72E-03 |
5.19E-03 |
4.62E-03 |
5.32E-03 |
|
|
||||||||
|
133 |
microg/L |
1147.42 |
1597.77 |
Wt % |
|
|
|
|
|
|
||||||
|
134 |
microg/L |
Wt % |
|
|
|
|
8.41E-04 |
7.75E-04 |
||||||||
|
135 |
microg/L |
103.51 |
108.29 |
Wt % |
8.84E-04 |
9.50E-04 |
8.66E-04 |
9.73E-04 |
8.18E-04 |
7.95E-04 |
||||||
|
136 |
microg/L |
Wt % |
6.37E-04 |
8.26E-04 |
6.24E-04 |
8.46E-04 |
4.34E-03 |
4.65E-03 |
||||||||
|
137 |
microg/L |
351.37 |
367.53 |
Wt % |
3.40E-03 |
3.72E-03 |
3.33E-03 |
3.81E-03 |
1.02E-02 |
1.02E-02 |
||||||
|
138 |
microg/L |
3.05 |
Wt % |
8.07E-03 |
9.09E-03 |
7.91E-03 |
9.31E-03 |
1.50E-03 |
1.28E-03 |
|||||||
|
139 |
microg/L |
Wt % |
1.41E-03 |
6.60E-04 |
1.38E-03 |
6.76E-04 |
5.74E-03 |
5.40E-03 |
||||||||
|
140 |
microg/L |
Wt % |
4.74E-03 |
1.97E-03 |
4.65E-03 |
2.02E-03 |
1.13E-03 |
9.03E-04 |
||||||||
|
141 |
microg/L |
0.37 |
Wt % |
9.67E-04 |
3.81E-04 |
9.47E-04 |
3.90E-04 |
1.60E-03 |
1.46E-03 |
|||||||
|
142 |
microg/L |
0.09 |
0.44 |
Wt % |
1.41E-03 |
7.21E-04 |
1.38E-03 |
7.38E-04 |
1.05E-03 |
8.54E-04 |
||||||
|
143 |
microg/L |
Wt % |
1.03E-03 |
4.27E-04 |
1.01E-03 |
4.37E-04 |
1.33E-03 |
9.83E-04 |
||||||||
|
144 |
microg/L |
Wt % |
1.14E-03 |
4.22E-04 |
1.12E-03 |
4.32E-04 |
7.77E-04 |
5.69E-04 |
||||||||
|
145 |
microg/L |
Wt % |
8.05E-04 |
2.24E-04 |
7.89E-04 |
2.29E-04 |
5.83E-04 |
4.81E-04 |
||||||||
|
146 |
microg/L |
Wt % |
6.87E-04 |
2.26E-04 |
6.73E-04 |
2.31E-04 |
3.99E-04 |
3.76E-04 |
||||||||
|
147 |
microg/L |
Wt % |
4.58E-04 |
|
4.49E-04 |
|
4.26E-04 |
3.64E-04 |
||||||||
|
148 |
microg/L |
Wt % |
4.43E-04 |
|
4.34E-04 |
|
2.27E-03 |
2.76E-03 |
||||||||
|
149 |
microg/L |
1.57 |
Wt % |
6.65E-04 |
1.17E-03 |
6.51E-04 |
1.20E-03 |
7.58E-04 |
7.90E-04 |
|||||||
|
150 |
microg/L |
0.32 |
Wt % |
5.31E-04 |
4.15E-04 |
5.20E-04 |
4.25E-04 |
|
|
|||||||
|
151 |
microg/L |
Wt % |
|
|
|
|
|
|
||||||||
|
152 |
microg/L |
Wt % |
1.17E-04 |
|
1.14E-04 |
|
|
|
||||||||
|
153 |
microg/L |
Wt % |
|
|
|
|
|
|
||||||||
|
154 |
microg/L |
0.13 |
0.19 |
Wt % |
1.00E-04 |
2.12E-05 |
9.81E-05 |
2.17E-05 |
1.57E-04 |
1.19E-04 |
||||||
|
155 |
microg/L |
Wt % |
|
|
|
|
3.55E-04 |
3.67E-04 |
||||||||
|
156 |
microg/L |
Wt % |
2.81E-04 |
|
2.76E-04 |
|
1.26E-04 |
1.10E-04 |
||||||||
|
157 |
microg/L |
Wt % |
1.10E-04 |
|
1.07E-04 |
|
2.05E-04 |
1.71E-04 |
||||||||
|
158 |
microg/L |
Wt % |
1.26E-04 |
|
1.23E-04 |
|
2.53E-05 |
2.57E-05 |
||||||||
|
159 |
microg/L |
Wt % |
|
|
|
|
|
|
||||||||
|
160 |
microg/L |
Wt % |
1.24E-04 |
|
1.22E-04 |
|
|
|
||||||||
|
161 |
microg/L |
Wt % |
|
|
|
|
|
|
||||||||
|
162 |
microg/L |
Wt % |
|
|
|
|
|
|
||||||||
|
163 |
microg/L |
0.17 |
0.09 |
Wt % |
2.23E-05 |
2.12E-05 |
2.18E-05 |
2.17E-05 |
|
|
||||||
|
164 |
microg/L |
Wt % |
|
|
|
|
|
|
||||||||
|
165 |
microg/L |
Wt % |
|
|
||||||||||||
|
166 |
microg/L |
Wt % |
|
|
||||||||||||
|
167 |
microg/L |
Wt % |
|
|
||||||||||||
|
168 |
microg/L |
Wt % |
|
|
||||||||||||
|
169 |
microg/L |
Wt % |
|
|
||||||||||||
|
170 |
microg/L |
Wt % |
|
|
||||||||||||
|
171 |
microg/L |
0.21 |
0.23 |
Wt % |
1.01E-05 |
6.00E-06 |
9.90E-06 |
6.14E-06 |
8.04E-04 |
1.05E-03 |
||||||
|
172 |
microg/L |
Wt % |
1.28E-04 |
3.91E-04 |
1.25E-04 |
4.00E-04 |
3.00E-03 |
3.80E-03 |
||||||||
|
173 |
microg/L |
0.76 |
1.74 |
Wt % |
6.63E-04 |
1.11E-03 |
6.50E-04 |
1.14E-03 |
|
|
||||||
|
174 |
microg/L |
Wt % |
|
|
||||||||||||
|
175 |
microg/L |
Wt % |
|
|
||||||||||||
|
176 |
microg/L |
Wt % |
|
|
|
|
|
|
||||||||
|
177 |
microg/L |
Wt % |
|
|
|
|
|
|
||||||||
|
178 |
microg/L |
Wt % |
2.08E-04 |
|
2.04E-04 |
|
|
|
||||||||
|
179 |
microg/L |
Wt % |
1.35E-04 |
|
1.32E-04 |
|
|
|
||||||||
|
180 |
microg/L |
Wt % |
3.39E-04 |
|
3.32E-04 |
|
|
8.68E-05 |
||||||||
|
181 |
microg/L |
Wt % |
1.73E-04 |
|
1.69E-04 |
|
|
2.38E-04 |
||||||||
|
182 |
microg/L |
22.05 |
23.14 |
Wt % |
|
2.07E-04 |
|
2.12E-04 |
|
|
||||||
|
183 |
microg/L |
13.86 |
12.99 |
Wt % |
|
|
|
|
2.26E-04 |
2.66E-04 |
||||||
|
184 |
microg/L |
25.50 |
25.94 |
Wt % |
2.55E-04 |
2.14E-04 |
2.49E-04 |
2.19E-04 |
|
|
||||||
|
185 |
microg/L |
Wt % |
|
|
|
|
2.07E-04 |
2.51E-04 |
||||||||
|
186 |
microg/L |
24.31 |
24.69 |
Wt % |
1.58E-04 |
1.87E-04 |
1.55E-04 |
1.91E-04 |
|
|
||||||
|
187 |
microg/L |
Wt % |
|
|
||||||||||||
|
188 |
microg/L |
Wt % |
|
|
||||||||||||
|
189 |
microg/L |
Wt % |
|
|
||||||||||||
|
190 |
microg/L |
Wt % |
|
|
||||||||||||
|
191 |
microg/L |
Wt % |
|
|
||||||||||||
|
192 |
microg/L |
Wt % |
|
|
||||||||||||
|
193 |
microg/L |
Wt % |
|
|
||||||||||||
|
194 |
microg/L |
Wt % |
|
|
||||||||||||
|
195 |
microg/L |
Wt % |
|
|
||||||||||||
|
196 |
microg/L |
4.89 |
4.51 |
Wt % |
|
|
||||||||||
|
197 |
microg/L |
2.73 |
3.05 |
Wt % |
|
|
||||||||||
|
198 |
microg/L |
197.94 |
202.87 |
Wt % |
|
|
||||||||||
|
199 |
microg/L |
325.93 |
329.40 |
Wt % |
4.45E-04 |
|
4.36E-04 |
|
9.40E-05 |
9.82E-05 |
||||||
|
200 |
microg/L |
453.21 |
446.66 |
Wt % |
6.62E-04 |
5.26E-04 |
6.49E-04 |
5.39E-04 |
|
|
||||||
|
201 |
microg/L |
251.25 |
257.33 |
Wt % |
4.38E-04 |
2.25E-04 |
4.29E-04 |
2.31E-04 |
|
|
||||||
|
202 |
microg/L |
571.27 |
559.62 |
Wt % |
7.50E-04 |
7.46E-04 |
7.35E-04 |
7.63E-04 |
|
|
||||||
|
203 |
microg/L |
1.47 |
1.34 |
Wt % |
1.70E-04 |
8.50E-05 |
1.66E-04 |
8.70E-05 |
5.10E-04 |
6.44E-04 |
||||||
|
204 |
microg/L |
187.65 |
195.41 |
Wt % |
5.83E-04 |
6.88E-04 |
5.72E-04 |
7.05E-04 |
|
|
||||||
|
205 |
microg/L |
Wt % |
3.35E-03 |
3.21E-03 |
3.28E-03 |
3.29E-03 |
7.03E-03 |
7.27E-03 |
||||||||
|
206 |
microg/L |
3.30 |
13.50 |
Wt % |
4.46E-03 |
5.18E-03 |
4.37E-03 |
5.31E-03 |
7.12E-03 |
7.02E-03 |
||||||
|
207 |
microg/L |
16.54 |
Wt % |
4.62E-03 |
5.23E-03 |
4.53E-03 |
5.35E-03 |
1.60E-02 |
1.66E-02 |
|||||||
|
208 |
microg/L |
7.36 |
25.58 |
Wt % |
9.78E-03 |
1.06E-02 |
9.59E-03 |
1.09E-02 |
|
|
||||||
|
209 |
microg/L |
415480 |
425899 |
Wt % |
4.93E+01 |
4.78E+01 |
0.00E+00 |
0.00E+00 |
|
|
||||||
|
230 |
microg/L |
Wt % |
|
|
0.00E+00 |
0.00E+00 |
|
|
||||||||
|
231 |
microg/L |
Wt % |
|
|
0.00E+00 |
0.00E+00 |
|
|
||||||||
|
232 |
microg/L |
5.45 |
3.91 |
Wt % |
2.95E-02 |
2.84E-02 |
1.45E-02 |
1.42E-02 |
2.28E-02 |
3.46E-02 |
||||||
|
233 |
microg/L |
Wt % |
|
|
|
|
|
|
||||||||
|
234 |
microg/L |
10.13 |
10.20 |
Wt % |
4.85E-04 |
4.61E-04 |
|
4.65E-04 |
6.37E-04 |
7.40E-04 |
||||||
|
235 |
microg/L |
17.14 |
20.52 |
Wt % |
1.07E-02 |
1.06E-02 |
8.84E-03 |
9.68E-03 |
1.45E-02 |
1.62E-02 |
||||||
|
236 |
microg/L |
Wt % |
8.41E-04 |
8.45E-04 |
5.34E-04 |
7.41E-04 |
9.19E-04 |
9.90E-04 |
||||||||
|
237 |
microg/L |
4.43 |
Wt % |
1.18E-03 |
1.17E-03 |
8.02E-04 |
1.17E-03 |
1.28E-03 |
1.61E-03 |
|||||||
|
238 |
microg/L |
1764.63 |
2085.38 |
Wt % |
1.67E+00 |
1.68E+00 |
1.37E+00 |
1.59E+00 |
2.24E+00 |
2.57E+00 |
||||||
|
239 |
microg/L |
13.55 |
11.79 |
Wt % |
4.34E-03 |
4.60E-03 |
4.27E-03 |
4.48E-03 |
6.32E-03 |
7.19E-03 |
||||||
|
240 |
microg/L |
Wt % |
4.79E-04 |
4.37E-04 |
4.34E-04 |
|
5.20E-04 |
7.64E-04 |
||||||||
|
241 |
microg/L |
Wt % |
|
|
|
|
||||||||||
|
242 |
microg/L |
Wt % |
|
|
|
|
||||||||||
|
243 |
microg/L |
Wt % |
|
|
|
|
||||||||||
|
244 |
microg/L |
Wt % |
|
|
|
|
||||||||||
|
245 |
microg/L |
Wt % |
|
|
|
|
||||||||||
|
246 |
microg/L |
Wt % |
|
|
|
|
||||||||||
|
247 |
microg/L |
Wt % |
|
|
|
|
||||||||||
References