
WSRC-TR-2000-00341
Entrained Solids, Strontium-Transuranic Precipitation,
and Crossflow Filtration of AN102 Small C
Charles A. Nash, Scott W. Rosencrance, and Bill R. Wilmarth
Westinghouse Savannah River Company
Aiken, SC 29808
Keywords: Hanford River Protection Project , Filtration, Separation Processes, Transuranic
This report was prepared as an account of work sponsored by an agency of the United States Government. Neither the United States Government nor any agency thereof, nor any of their employees, makes any warranty, express or implied, or assumes any legal liability or responsibility for the accuracy, completeness, or usefulness of any information, apparatus, product or process disclosed, or represents that its use would not infringe privately owned rights. Reference herein to any specific commercial product, process or service by trade name, trademark, manufacturer, or otherwise does not necessarily constitute or imply its endorsement, recommendation, or favoring by the United States Government or any agency thereof. The views and opinions of authors expressed herein do not necessarily state or reflect those of the United States Government or any agency thereof.
This report has been reproduced directly from the best available copy.
Available for sale to the public, in paper, from: U.S. Department of Commerce, National Technical Information Service, 5285 Port Royal Road, Springfield, VA 22161, phone: (800) 553-6847, fax: (703) 605-6900, email: orders@ntis.fedworld.gov online ordering: http://www.ntis.gov/support/ordering.htm
Available electronically at http://www.osti.gov/bridge/
Available for a processing fee to U.S. Department of Energy and its contractors, in paper, from: U.S. Department of Energy, Office of Scientific and Technical Information, P.O. Box 62, Oak Ridge, TN 37831-0062, phone: (865 ) 576-8401, fax: (865) 576-5728, email: reports@adonis.osti.gov
Summary
A crossflow filtration campaign to remove entrained solids from "Small" Envelope C was completed. A 2-foot long, 3/8" internal diameter, 0.1 micron pore size Mott crossflow filter tube was used. An Envelope C (241-AN-102) sample containing entrained solids was then successfully pretreated for removal of strontium-90 and transuranic activity. The 1.2-liter sample was caustic adjusted, strontium and permanganate precipitated, and crossflow filtered in a bench scale demonstration. Filtration fluxes for the precipitate ranged from 0.05 to 0.15 gpm/ft*ft for a slurry with an insoluble solids content of ~1.5 wt%. Transmembrane pressures were in the range of 30 to 70 psid and crossflow velocities were in the range of 9 to 15 ft/s. This test with the radioactive waste sample confirms filter flux results obtained using a strontium-transuranic precipitation simulant that was conducted with a pilot-scale crossflow ultrafiltration unit. In that simulant test, the initial 2-hour filter flux ranged from 0.065 to 0.080 gpm/ft2 at a transmembrane pressure of 52 psid and a crossflow velocity of ~12 ft/sec.
The filtrate product was decontaminated for strontium by a factor of 30 (1.7 uCi/ml at 5.8 M sodium). This work provides important confirmation of the new process to achieve both acceptable filterability and decontamination for Envelope C wastes to be treated by the Hanford River Protection Project.
Introduction
This work is the first active crossflow filter testing of the new strontium-permanganate process for Envelope C at SRTC. Extreme filtration difficulties with the ferric hydroxide precipitation led to investigations of other chemistries with simulants. The current process includes the isotopic dilution by precipitation from inactive strontium nitrate addition. A permanganate strike to replace ferric reagents was found to provide practical filterability and good lanthanide or transuranic decontamination. That work had been supported with simulant and active beaker testing.
A detailed literature background supporting the new process is given in the second reference. Water treatment as well as testing of nuclear waste treatment with permanganate has been studied in the past. However, adaptation of the process to Hanford River Protection Project (RPP) needs is demonstrated in this report. The precipitation recipe used here was the result of extensive optimization work at SRTC.
In addition to the chemistry modification for transuranic removal, the prefiltration step to remove entrained solids before precipitation was removed from the original flowsheet. This strategy had several benefits. First, the solids activities were found to put them in the High Level Waste category. Their inclusion with the precipitate simplifies process flow. Secondly, initial crossflow testing at PNNL and at SRTC found that the entrained solids alone are very difficult to filter. Combination of them with the precipitate created by permanganate has now been shown to make them easier to filter. SRTC personnel have also conducted a pilot-scale demonstration of the strontium-manganese precipitation process and crossflow filtration using an Envelope C (tank 241-AN-107) simulant. Together, the small-scale active and pilot-scale simulant process test results confirm the design basis for the strontium-transuranic removal process planned for the RPP Waste Treatment Plant (WTP). Specifically, an average filter flux rate of 0.065 gpm/ft2 is achievable for filtration of the strontium-manganese precipitate. Strontium-90 and transuranic element removal averages are greater than 80 and 90%, respectively.
Apparatus
Crossflow filtration was performed with a Cells Unit Filter (CUF) rig that was set up in Cell 16, B-Block, at SRTC. Figure 1 shows the unit without cooling tubes connected to the heat exchanger for clarity. Feed from the reservoir at the left goes to a progressive cavity pump. The pump is operated at variable speed by controlling air pressure to the air motor that drives it. Liquid is pumped through a magnetic flowmeter and heat exchanger that removes pump heat. It then passes down the center of a crossflow filter of 2-foot porous length. A throttle valve downstream drops fluid pressure back to atmospheric.
Figure 1. Photograph of the Cells Filter Unit in a Nonradioactive Laboratory

Filtrate flowrate was measured with a sightglass and stopwatch. The simple backpulse system is manually operated. The backpulse chamber is first charged with filtrate followed by compressed air. Quickly opening a toggle valve below the chamber forces reverse flow of filtrate upon the filter medium. Standard Bourdon-type pressure gauges indicate pressure. A thermocouple mounted near the bottom of the reservoir measures slurry temperature directly. Details of the CUF are documented on six approved engineering drawings.
The filter in this work was a 3/8-inch internal diameter, 2-foot long Mott Metallurgical sintered stainless steel filter. The nominal pore size was 0.1 micron. The single filter tube was mounted in a stainless steel housing of welded construction.
Experimental
Tank 241-AN-102 Sample History
The Hanford site contractor obtained approximately 16.5-liters of waste solution from tank 241-AN-102 during 1998. These samples were obtained by lowering sample bottles into the liquid phase of the waste. These samples are denoted as "grab samples", since the contents of tank 241-AN-102 was not agitated prior to or during the sampling event. The Hanford site contractor packaged samples obtained from tank 241-AN-102 for shipment to the Savannah River Technology Center. The Savannah River Technology Center received as five separate shipments from October 1998 through December 1998 approximately 16.5 liters of waste obtained from tank 241-AN-102 at the Hanford site.
The current work was performed using material from a grand composite of all the 241-AN-102 material that was received for Part B1. Thirty-two samples were composited, characterized, and a portion was diluted to 7 M sodium for this work. Summary information on the samples is shown in Table 1.
Table 1. Sample Identifications for Small C Composite
|
Number of Jars |
Jar Label |
Sample Date |
Sample Type |
|
6 |
2AN 98-43 to |
07/21/98 |
Unaltered Grab Sample |
|
6 |
2AN 98-49 to |
07/22/98 |
Unaltered Grab Sample |
|
5 |
2AN 98-58 to |
08/10/98 |
Unaltered Grab Sample |
|
5 |
2AN 98-63 to |
08/11/98 |
Unaltered Grab Sample |
|
5 |
2AN 98-68 to |
08/12/98 |
Unaltered Grab Sample |
|
5 |
2AN 98-LCOMP1,3,4,5,8 |
07/21/98 |
Liquid Core Composite |
Filter Operation
General operation of the CUF follows an SRTC procedure. Activities and data are recorded in a controlled laboratory notebook. The rig internals are first rinsed with flush solutions or DI water per the steps below. The filter cleaning fluids were prefiltered with 0.22 micron nylon filters before use.
A 0.1 micron-pore Mott filter tube manufactured for "liquid" service was used in this work. The cold CUF work (ref. 1) found that Mott makes different sintered 0.1 micron stainless steel for "liquid" and "gas" service and that the "liquid" style is a requirement for our work. The filter has a two-foot active length, 3/8" ID bore, and 1/16" wall thickness.
Clean water fluxes were taken after the rig was flushed with cleaning fluids. Transmembrane pressures were between 5 and 20 psid and fluxes were measured for periods of 20 minutes or more per the TTP, after initial backpulsing. The purpose of cleaning the filter rig and obtaining clean water flux measurements is to ensure the rig is cleaned and establish a baseline filter flux to determine if filter fouling occurs during tests with the waste sample.
Before the 241-AN-102 sample with entrained solids or slurry was run the rig was drained completely of water using a vacuum flask attached to the pump drain. The pump drain is the lowest point in the system. Vacuum was supplied by squeezing a suction bulb with a manipulator.
The precipitation of a 1.2 liter subsample of the 241-AN-102 waste sample (identified as "Small C") followed a directive from the customer. The material provided to the precipitation/filtration campaign was 7 M in sodium. Addition of the solution volumes in the directive yields the following for 1.2 liters of 7 M sodium feed:
Since the feed has several tenths of a molar free hydroxide the final free hydroxide in the batch is probably 0.9 M.
The following steps cover dilution to approximately 6 M sodium, caustic adjustment, and precipitation. Actual volumes as measured were within 0.05 ml of the ideal targets.
Operation of the CUF involved the following routine:
Results and Discussion
Filtration – Entrained Solids
In the first operation of the Filter Unit with actual tank material, Small C entrained solids were found to filter with much difficulty. There was no precipitation done here.
Clean water fluxes were first obtained after the unit was installed in Cell 16 of SRTC B-block. Figure 2 below summarizes the data obtained. The filter was judged to be clean because the fluxes were an order of magnitude above what would be expected for slurry filtration at similar transmembrane pressures. Mott does not publish a graph of clean water flux for this filter as they do for some of their other sintered stainless steel products.
Figure 2. Mott 0.1 Micron 2-Foot Filter Clean Water Fluxes
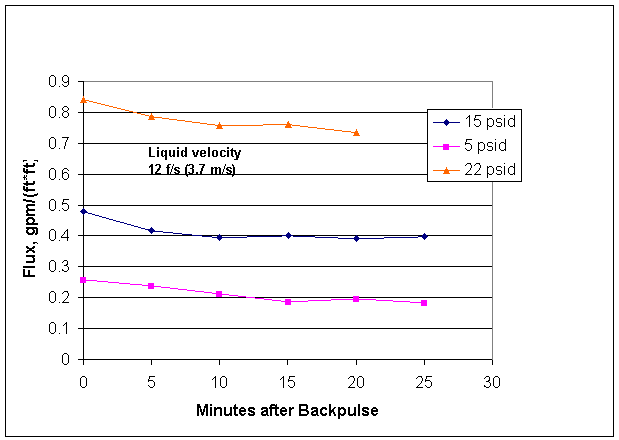
The temperature for the Figure 2 data was 28 Celsius on average, where 25 +/- 5 Celsius was specified.
Figure 3 below shows how filtration of the Small C sample with entrained solids (equal or less than 0.1 wt%) was indeed much smaller than clean water fluxes.13 Consecutive run conditions were all at 29 to 30 Celsius except for the fifth run where it was controlled at 45 Celsius for the sake of learning what effect higher temperature might have. This higher temperature value was easily provided by controlling the removal of pump heat from the loop.
Transmembrane pressure and velocity conditions used in Figure 3 are covered by Table 2. They were provided in the customer-provided test specification, reference 9. Crossflow velocity and transmembrane pressure ranges are consistent with past work on the crossflow filtration of metal oxide sludges, though a higher transmembrane pressure range to 70 psid was explored here as well (past testing upper pressures were typically 45 psid).,
Table 2. Consecutive Set of Conditions for Entrained Solids Filtration
|
Run Order |
TMP, psid |
Velocity, ft/s(m/s) |
|
Run Order |
TMP, |
Velocity, ft/s(m/s) |
|
1 |
50 |
12 (3.7) |
4 |
70 |
12 (3.7) |
|
|
2 |
30 |
15.3 (4.7) |
5 (45 C) |
50 |
12 (3.7) |
|
|
3 |
70 |
9 (2.7) |
6 |
50 |
12 (3.7) |
Figure 3. Entrained Solids Filtration of Small C (AN102)
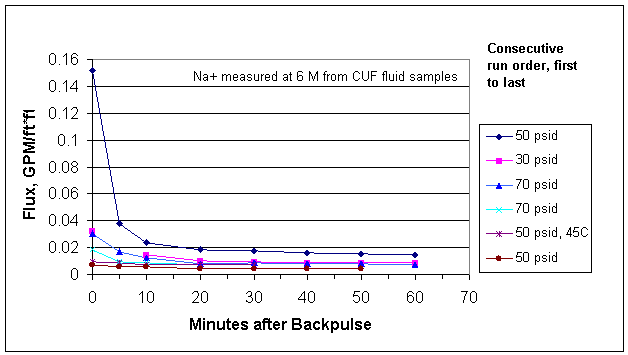
The average flux rapidly dropped below 0.05 gpm/ft*ft in the first run and remained low despite variations of flow, pressure and temperature conditions. Average flux for the consecutive runs are shown in Figure 4.
The flux averages for the entrained solids runs were analyzed using a statistical package (SAS Institute, Inc.) to determine a best linear fit flux model. Average flux, the dependent variable, was correlated against crossflow velocity, transmembrane pressure, and chronological run number (runtime, hours). Crossflow velocity and transmembrane pressure were found to be insignificant as would be surmised from the plots above.
Figure 4. Average Flux for Consecutive Small C Entrained Solids Runs
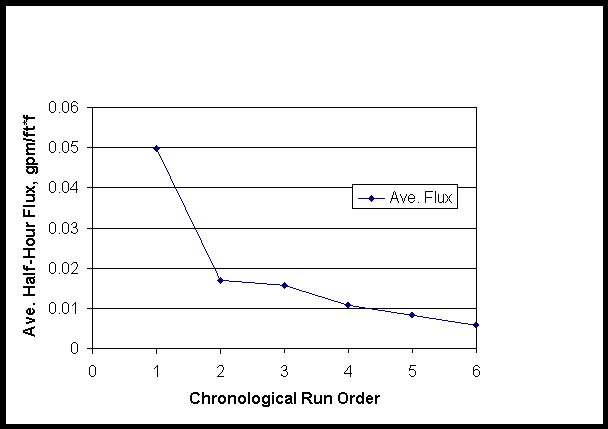
Runtime was significant and a meaningful linear model was:
Average flux (gpm, ft*ft) = (0.0431+/-0.0095) – (0.007+/-0.0024)*(runtime, hours)
The linear coefficients have standard estimates of error given. The time effect could be either from entrained fines fouling the filter or from their shear degradation.
Filtration – Strontium/TRU Precipitation
It is clear from Figure 4 that a process modification was needed to boost throughput to achieve the WTP design basis average filter flux of 0.065 gpm/ft2. It was decided to run the Small C (241-AN-102) precipitation process with entrained solids included. One purpose would be to see if the strontium/permanganate slurry would act as a filter aid for the entrained solids. The entrained solids present in the 241-AN-102 waste sample are considered high-level waste based on TRU content (23.5 mCi/gm) and Sr-90 content (1.16E+03 mCi/gm). Jointly separating the entrained solids with the Sr-Mn precipitate would simplify the process as well by eliminating the entrained solids filtration step for Envelope C. One technical risk was that the presence of entrained solids in the slurry might impact decontamination. A goal of this work was therefore to see if leaving the entrained solids in with the precipitate still provides acceptable decontamination.
1200 ml of Small C (AN102) at 7 M sodium was diluted to ~6 M sodium and precipitated per the Experimental section discussed above. The batch volume was about 1640 ml after all additions were made to the solution. The slurry was held at 50 Celsius for 4 hours and then introduced very slowly into the Filter Unit, allowing the heat exchanger to cool the material immediately while it flowed into the concentrate loop. The slurry was filtered at 25 +/- 5 oC The conditions for filtration are shown in Table 3 and are taken from the customer’s test specification.9 An average analysis of the slurry is in Appendix A.
Table 3. Consecutive Sets of Conditions for Sr/Mn Precipitate Filtration
|
Run Order |
TMP, psid |
Velocity, ft/s(m/s) |
Run Order |
TMP, psid |
Velocity, ft/s(m/s) |
|
|
1 |
50 |
12 (3.7) |
5 |
50 |
12 (3.7) |
|
|
2 |
30 |
9 (2.7) |
6(45 C) |
70 |
9 (2.7) |
|
|
3 |
30 |
15.3 (4.7) |
7 |
30 |
9 (2.7) |
|
|
4 |
70 |
9 (2.7) |
8 |
30 |
15.3 (4.7) |
Figure 5. Flux Data vs. Time for First Entrained Solids and All Slurry Runs
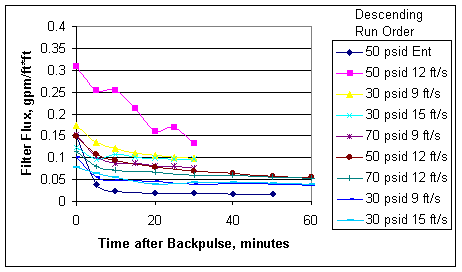
Figure 5 above shows the first (highest flux) entrained solids filtration run data compared with all eight of the precipitate slurry data. The slurry data are presented consecutively. It is clear that the precipitate acts like a filter aid and masks the filter fouling that entrained solids alone will cause. The data also show that the flux from slurry filtration is consistently above 0.04 gpm/ft*ft and that backpulsing can increase it above 0.1 gpm/ft*ft.
Both Figure 5 and 6 show how filter flux is mostly a function of run time rather than conditions. A linear statistical analysis of the data also shows this to be true. Runtime was significant and a meaningful linear model was:
Average flux (gpm, ft*ft) = (0.14+/-0.022) – (0.0109+/-0.00169)*(runtime, hours)
The linear coefficients have standard estimates of error as shown. The first run was omitted because it was so much higher than following runs. Nonlinearity it introduced would not have led to a statistically significant linear model.
Modeling the final flux point in each run rather than using run average fluxes also revealed no statistical significance of varying the transmembrane pressure and crossflow velocity. The result for the linear fit was:
Average flux (gpm, ft*ft) = (0.13+/-0.008) – (0.0132+/-0.00153)*(runtime, hours)
The similarity of this equation with the first one shows that the data cannot support statistical significance of filter conditions versus simple runtime. It is clear that slurry particle changes from hours of pump shearing are the main influence on filter flux. Use of low shear pumps and throttle valves in plant design may thus slow the rate of flux reduction. Reduction of tank heels between precipitate batches may also help. Effects of differing weight percent and mixing of slurry with different histories were not evaluated here because the same slurry batch was used in all filter runs.
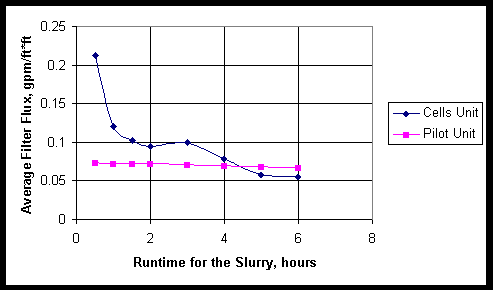
These results with the small C sample compare favorably and support pilot-scale test results for the Sr-Mn precipitated 241-AN107 simulant where the average filter flux was found also to be related to run time by the following data fit:
Average filter flux (gpm/ft2) = 0.0743e-0.0151*T(hours)
The pilot-scale simulant test results are valid for a 2wt% insoluble solids slurry run at ~12 ft/sec and a transmembrane pressure of ~52 psid. The small C filter flux is ~0.09gpm/ft2 whereas the pilot-scale simulant filter flux is ~0.07gpm/ft2 after a 5-hour run time. The small C filter flux is ~0.03gpm/ft2 whereas the pilot-scale simulant filter flux is ~0.06gpm/ft2 after a 10-hour run time. Figure 7 shows the above pilot scale equation with data points from the small C filtration campaign. This indicates the pilot-scale simulant test results conservatively under estimate filter flux for several hours without filter back-pulse or cleaning. For a run duration longer than 5-hours, the pilot-scale simulant results do not conservatively estimate the filter flux compared to the results obtained with the Small C waste sample.
Shear degradation of the slurry particles probably played a role in the flux degradation. Appendix B shows particle size data for three slurry samples taken from the loop. The first sample shows a bimodal particle size. It is fresh precipitate slurry after only momentary pumping in the filter unit (enough to mix the loop and draw a sample). The mean particle diameter was 40 microns. The two other samples were taken after 5 and 7 hours of running. They show that mean particle size has settled to a value around 10 microns, and that the bimodal distribution has now merged to a single distribution.
Figure 6. Average Filter Flux vs. Consecutive Run Order for ~1.5 wt% Slurry
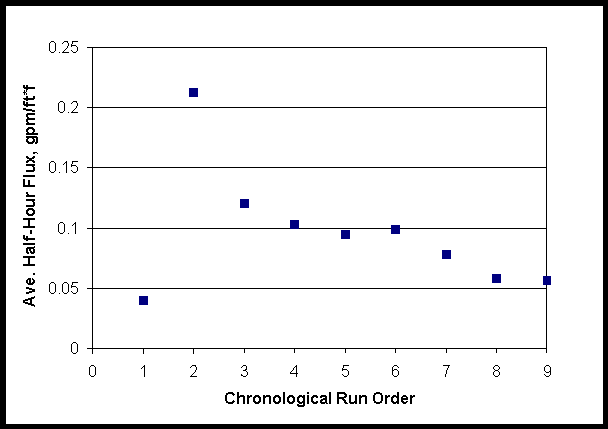
Figure 7. Comparison of Cells Filter and Pilot Filter Data
Component Separation and Decontamination Data - Entrained Solids
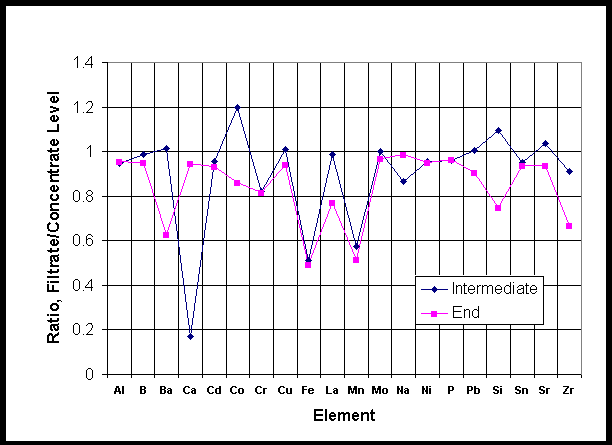
This section presents component separation data observed when the Small C sample before precipitation was passed through the crossflow filter. Figures 8 and 9 show anion and elemental analyses of the samples of concentrate and filtrate from the entrained solids runs. The initial feed had been sampled as well as pairs of concentrate and filtrate after the first day of running (3 runs) and the second day (another 3 runs). Samples were analyzed in duplicate. Analyses are reported in Appendix C.
Anions show no significant separation as expected. Figure 8 also shows uranium concentration on both sides of the filter to be equal. The uranium is thus shown to be a soluble species.
Figure 9 as well shows that most elements in AN102 are associated with soluble species and not with the entrained solids. Interestingly, though, the iron and manganese levels do show a difference, suggesting that these are the main components of the entrained solids. While these elements are among the major ones found in tank 241-AN-102 sludge, one would also expect aluminum to be found. , Calcium and chromium are also likely constituents of sludge. It is unfortunate that the latest 241-AN-102 sludge sampling analysis did not quantify the amounts of iron or manganese to compare with the aluminum and sodium that were found.
Figure 8. Degree of Filtration - Species in the Entrained Solids Sample
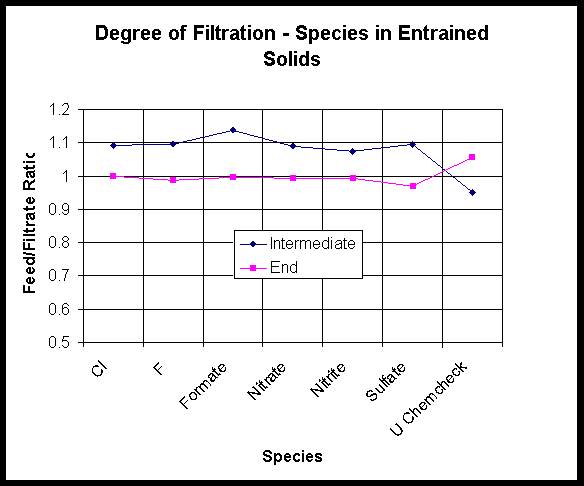
Figure 9. Elemental Split across the Filter - ICP-ES Data
Component Separation and Decontamination Data Precipitate Slurry
Tables 4 and 5 below show how the precipitation process removed various elements, both total (Table 4) and active (Table 5). The major elements of interest have been shaded in Table 4. "Slurry Based" data correspond to comparisons where elemental split between concentrate and slurry samples are made. There is no dilution correction because the samples are taken at the same time. "Feed Based" values compare removals in the filtrate with the initial feed (defined as 241-AN-102 sample after dilution and addition of chemical reagents) and a 10% dilution correction was applied based on sodium level measurements.
"Filt-A" and "Filt-B" in Table 4 are data from samples taken after 3 hours of running the CUF loop and obtaining filtration data. "Filt-Product" data are from a composite product sample taken at the end of the filtration campaign.
The main elements removed by the process are strontium-90 and total iron. A feed-based iron removal DF of 5 was seen in the initial simulant work with an 241-AN-107 simulant, and this compares well with the current work.2 The fact that the slurry-based strontium and Sr-90 (active strontium) DF’s compare well between the two tables shows that isotopic dilution goes most of the way to completion as desired. The fact that the feed-based strontium is unchanged by the process shows that the 241-AN-102 material is probably saturated in total strontium.
Aluminum and nickel are not removed here, similar to the simulant work. Calcium is weakly removed here (DF of 1.5) compared with a DF of 2.4 for the simulant work.
Table 4. Decontamination Factors for the Sr/Permanganate Precipitation
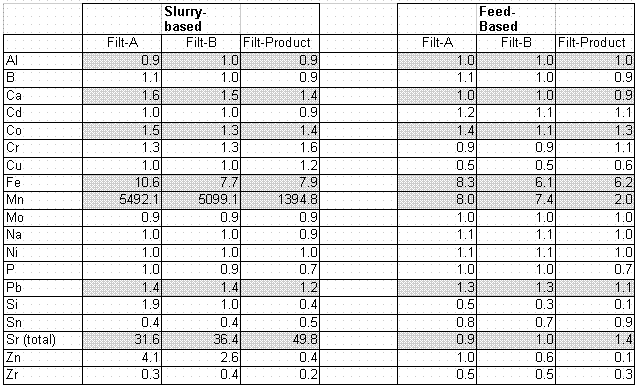
Slurry-based manganese is very high because this, like strontium, is an element added to the process. Most of the added manganese is retained in the slurry concentrate.
Cobalt is hardly removed at all as shown by the low total cobalt DF’s in Table 4 and the fact that the Co-60 measured in the "End filtrate" was 0.058 uCi/ml, and the total Co-60 in the final filtrate product was 0.04 uCi/ml at a measured sodium level of 5.8 M.
Table 5 confirms the expectation that cesium is not removed by the process. Curium is slightly more difficult than Americium to remove.
Table 5. Feed-Based Decontaminations for the Sr/Permanganate Precipitation
|
DF |
Cs-137 |
Sr-90 |
Am-241 |
Cm-244 |
|
Intermediate Filtrate |
1.1 |
28 |
8.3 |
5.1 |
|
End Filtrate |
1.1 |
33 |
7.6 |
4.9 |
|
Filtrate Product |
1.0 |
68 |
9.2 |
7.2 |
After the entrained solids filtration campaign and the precipitate filtration campaign, the rig was drained and flushed with one inventory volume of inhibited water (DI water with 0.01 M NaOH added) followed by a flush with 1 M nitric acid and a second water flush. The result after both cleaning series was that the final washwater was less then 3 percent of the activity of the respective concentrate that had been in the rig. Figure 9 shows the levels of nuclides found in the fluid after each step.
Compositions of the acid and final flush water solutions from the filter rig are found in Appendix D. In both the cases of cleaning after entrained solids and precipitate slurry the activity of the last flush is 3% or less of initial activity in the loop during operation. The second table in the appendix shows how extra strontium and manganese is present as leftovers from the precipitate filtration.
Figure 10. Washout of Nuclides after Entrained Solids Filtration
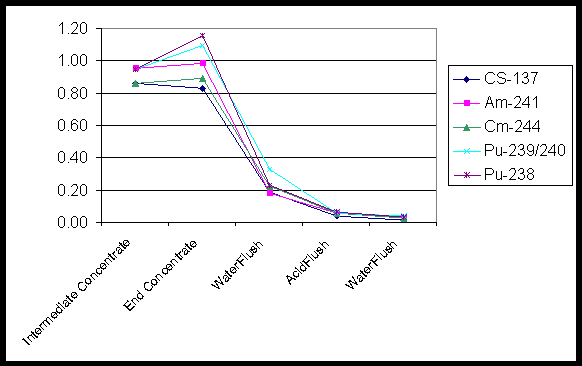
Table 6. Clean Water Fluxes After Final Flushing
|
Transmembrane Pressure, psid |
Average Water Flux, gpm/ft*ft |
|
10 |
0.14 |
|
15 |
0.21 |
|
20 |
0.35 |
Final clean water fluxes were about half of those of a new filter (Table 6). The cleaning was successful in recovering fluxes much higher than those seen in the slurry filtrations.
Conclusions
Entrained solids filtration is very difficult at 30 and 45 Celsius and under the range of transmembrane pressures 30 to 70 psid and crossflow velocities in the range of 9 to 15 ft/s. Filter flux was more a function of time than of set conditions. It is recommended that the entrained solids filtration step be combined with the Sr-Mn precipitation and filtration step for Envelope C. The current work shows that elimination of the entrained solids filtration step provides both practical filtration fluxes and acceptable decontamination in the precipitation/filtration process. Process simplification and avoidance of entrained solids filtration difficulties are thus both realized.
The strontium and permanganate process was run successfully on 1200 ml of 241-AN-102 feed after adjustment to 6 M sodium. Filtration fluxes exceeded 0.04 gpm/ft*ft in a process where the precipitate was formed on top of the entrained solids. The inclusion of entrained solids did not hinder the process removal of strontium-90 and alpha emitters (chiefly americium-241). Filter flux was governed by slurry pumping time and no statistical significance of transmembrane pressure and crossflow velocity could be supported from the data.
Cleaning the rig with inhibited water followed by 1 M nitric acid, and a final flush of inhibited water was effective in flushing the rig so that the final water flush had 3% or less of initial loop activity. Clean water flux was half that of a new filter but was significantly above process operating fluxes.
Recommendations for Further Work
Filter flux enhancement through the use of surfactants or filter aids is recommended. Filter flux rate as a function of solids loading is recommended, though a larger sample would be needed so that concentration of precipitate could be performed. Many chemical sensitivity, mechanism, post filtration solids formation and safety related studies are expected in the ongoing process development.
References
Appendix A: Analysis of the Small C Input Sample
The following table provides the average analysis from three samples of the small C liquid plus entrained solids used in the entrained solids campaign and used again in the precipitation campaign. Measurements on each sample were made in duplicate. The first sample was the small C sample (composite of 8 jars) after dilution to a target of 6 M sodium. The intermediate and end concentrate samples were drawn from the filter unit after some entrained solids filtration. It is seen that the samples show little variation as expected because the rig was clean when the Small C sample was introduced. The results also compare well with the initial and separate SRTC analysis to characterize C except that curium (possible contaminant) was found to be about a quarter of the characterization value.
|
Analytical Results For Small C |
||||||||||
|
Initial Concentrate |
Intermediate Conc. |
End Concentrate |
Dil. Corr. |
Dil. Corr. |
Percent |
Units |
||||
|
Method |
Analysis 1 |
Analysis 2 |
Analysis 1 |
Analysis 2 |
Analysis 1 |
Analysis 2 |
Average |
St. Dev. |
error |
|
|
Al |
332.37 |
333.75 |
296.02 |
292.162 |
309.1 |
297.4 |
9304 |
559 |
6.0 |
mg/liter |
|
B |
0.80 |
0.83 |
0.685 |
0.687 |
0.7 |
0.7 |
22 |
2 |
8.2 |
mg/liter |
|
Ba |
0.04 |
0.04 |
0.042 |
0.037 |
0.1 |
0.1 |
1 |
0 |
28.4 |
mg/liter |
|
Ca |
11.04 |
11.56 |
not avail. |
10.317 |
10.3 |
10.5 |
322 |
16 |
5.1 |
mg/liter |
|
Cd |
1.38 |
1.37 |
1.246 |
1.196 |
1.3 |
1.2 |
39 |
2 |
5.9 |
mg/liter |
|
Co |
0.14 |
0.13 |
0.152 |
0.113 |
0.2 |
0.1 |
4 |
1 |
14.7 |
mg/liter |
|
Cr |
6.17 |
6.13 |
5.694 |
5.548 |
6.1 |
5.7 |
176 |
8 |
4.6 |
mg/liter |
|
Cu |
0.61 |
0.60 |
0.546 |
0.526 |
0.6 |
0.5 |
17 |
1 |
6.5 |
mg/liter |
|
Fe |
1.05 |
1.05 |
1.06 |
1.054 |
1.3 |
1.2 |
33 |
3 |
7.9 |
mg/liter |
|
La |
0.35 |
0.38 |
0.292 |
0.315 |
0.4 |
0.3 |
10 |
1 |
11.1 |
mg/liter |
|
Li |
< 0.030 |
< 0.030 |
< 0.030 |
< 0.030 |
< 0.030 |
< 0.030 |
below det. |
below det. |
mg/liter |
|
|
Mn |
0.43 |
0.42 |
0.384 |
0.377 |
0.4 |
0.4 |
12 |
1 |
6.2 |
mg/liter |
|
Mo |
1.37 |
1.38 |
1.269 |
1.23 |
1.3 |
1.3 |
39 |
2 |
4.5 |
mg/liter |
|
Na |
5345.19 |
5049.34 |
4624.672 |
4773.494 |
4761.5 |
4356.6 |
144554 |
10278 |
7.1 |
mg/liter |
|
Ni |
9.24 |
9.40 |
8.332 |
8.227 |
8.8 |
8.4 |
262 |
15 |
5.7 |
mg/liter |
|
P |
44.66 |
42.81 |
39.212 |
36.569 |
40.5 |
38.6 |
1212 |
88 |
7.3 |
mg/liter |
|
Pb |
4.24 |
4.30 |
3.759 |
3.774 |
4.3 |
4.0 |
122 |
7 |
6.1 |
mg/liter |
|
Si |
0.81 |
0.77 |
0.693 |
0.612 |
1.0 |
0.8 |
24 |
4 |
18.3 |
mg/liter |
|
Sn |
0.67 |
0.70 |
0.662 |
0.701 |
0.8 |
0.7 |
21 |
1 |
5.8 |
mg/liter |
|
Sr |
0.08 |
0.08 |
0.072 |
0.067 |
0.1 |
0.1 |
2 |
0 |
7.4 |
mg/liter |
|
Ti |
< 0.030 |
< 0.030 |
< 0.030 |
< 0.030 |
0.0 |
< 0.030 |
below det. |
below det. |
mg/liter |
|
|
V |
0.06 |
0.05 |
0.046 |
< 0.040 |
0.1 |
< 0.040 |
2 |
1 |
30.8 |
mg/liter |
|
Zn |
< 0.030 |
< 0.030 |
< 0.030 |
< 0.030 |
< 0.030 |
< 0.030 |
below det. |
below det. |
mg/liter |
|
|
Zr |
0.27 |
0.20 |
0.245 |
0.17 |
0.3 |
0.2 |
7 |
1 |
21.1 |
mg/liter |
|
Sr-90 |
3.90E+06 |
3.20E+06 |
3.35E+06 |
3.50E+06 |
47.13 |
4.07 |
8.6 |
uCi/ml |
||
|
CS-137 |
2.12E+07 |
2.09E+07 |
1.84E+07 |
1.79E+07 |
1.75E+07 |
1.75E+07 |
255.62 |
23.00 |
9.0 |
uCi/ml |
|
Am-241 |
9880 |
9840 |
9660 |
9080 |
9810 |
9590 |
0.1303 |
0.0040 |
3.1 |
uCi/ml |
|
Cm-244 |
4290 |
4960 |
3720 |
4250 |
3910 |
4330 |
0.0573 |
0.0058 |
10.0 |
uCi/ml |
|
Pu-239/240 |
405 |
403 |
420 |
345 |
470 |
417 |
0.0055 |
0.0005 |
9.8 |
uCi/ml |
|
Pu-238 |
224 |
225 |
220 |
205 |
296 |
222 |
0.0031 |
0.0004 |
13.9 |
uCi/ml |
|
Sample 1 |
Sample 2 |
Sample 3 |
Average |
Standard |
Percent |
|||||
|
IC_Anions |
Analysis 1 |
Analysis 2 |
Analysis 1 |
Analysis 2 |
Analysis 1 |
Analysis 2 |
Molarity |
Deviation |
error |
|
|
mg/liter |
mg/liter |
mg/liter |
mg/liter |
mg/liter |
mg/liter |
M |
M |
% |
<<=units |
|
|
Cl |
65 |
68 |
59 |
59 |
58 |
59 |
0.052 |
0.003 |
6.7 |
|
|
F |
45 |
46 |
40 |
40 |
38 |
38 |
0.065 |
0.006 |
8.5 |
|
|
Formate |
234 |
235 |
208 |
192 |
189 |
189 |
0.139 |
0.015 |
10.5 |
|
|
Nitrate |
4140 |
4134 |
4693 |
4701 |
4685 |
4660 |
2.178 |
0.137 |
6.3 |
|
|
Nitrite |
1866 |
1892 |
1638 |
1651 |
1643 |
1633 |
1.122 |
0.080 |
7.2 |
|
|
Oxalate, Phosphate, and Magnesium were all below detection limits |
||||||||||
|
Sulfate |
280 |
294 |
254 |
257 |
257 |
251 |
0.083 |
0.005 |
6.5 |
|
|
TIC |
638.6 |
672.2 |
574 |
737 |
19664 |
2038 |
10.4 |
mg/liter |
||
|
TOC |
4335 |
3874 |
1397 |
1457 |
82973 |
46724 |
56.3 |
mg/liter |
||
|
U Chemch |
0.42 |
0.43 |
0.39 |
0.37 |
0.38 |
0.38 |
12 |
1 |
6.1 |
mg/liter |
|
Mass 238 |
0.65 |
0.609 |
0.518 |
0.49 |
17 |
2 |
13.3 |
mg/liter |
||
Appendix B: Particle Size Data
Concentrate slurry from the CUF filter loop was sampled at the beginning, after the fifth run, and at the end of the filter testing campaign. The three pages that follow show the volume percent particle size for the three slurry samples.
The following are explanations for the data on each page:
20%, 50%, 80% - Percentile points (microns) showing a given percent of the volume (or weight if the specific gravity for all the particles is the same; generally a good assumption) is smaller than the indicated size. The 50% number is the median diameter.
Mean Value is the mean diameter, microns, of the volume distribution. This is a weighted value of the "average particle size", or center of gravity of the distribution.
Mean Area is the mean diameter of the area distribution in microns. It is an "average particle size" which is inversely related to the Calculated Specific Area below.
Calculated Specific Area (square meters per ml) provides an indication of specific surface area. Since its computation assumes solid spherical particles it should not be interchanged with BET or other adsorption methods of surface determination since it does not include porosity or unique topographic characteristics of the particles.
Standard Deviation, microns, describes the width of the measured particle size distribution. It does not provide an indication of the statistical error about the mean of multiple measurements.
Standard Deviation = (84th minus 16th percentile)/2
Particles outside of the measuring range are not included in the distribution.
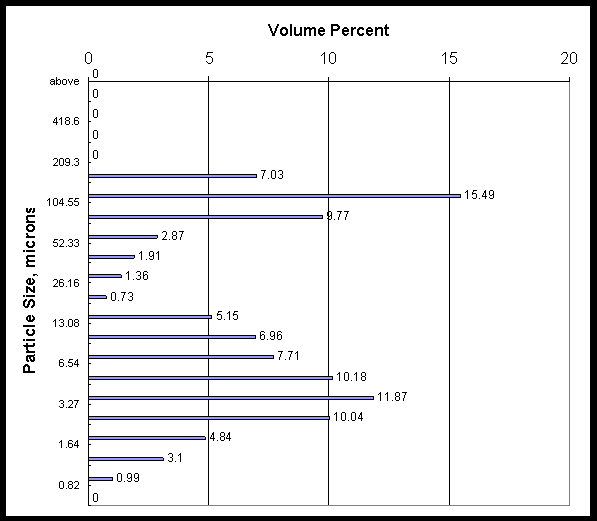
Data are the average of three measurements.
|
Mean value = 40 microns |
Mean Area = 5.03 microns |
|
20% is 2.85 microns |
Calculated Specific Area =1.192 m2/ml |
|
50% is 8.37 microns |
Standard Deviation = 50.42 microns |
|
80% is 93.94 microns |
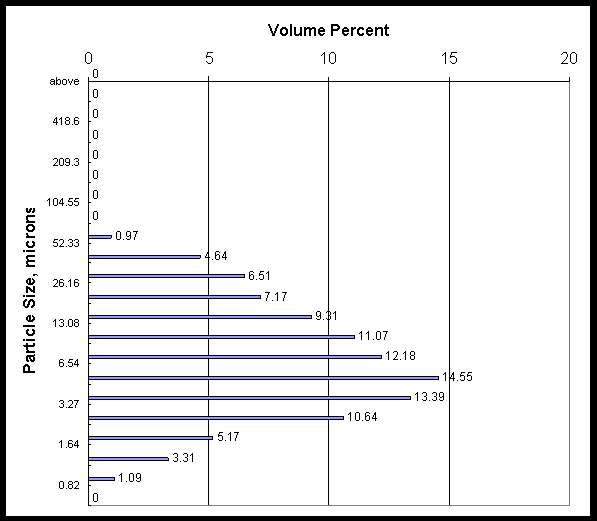
Data are the average of three measurements.
|
Mean value = 10 microns |
Mean Area = 4.21 microns |
|
20% is 2.73 microns |
Calculated Specific Area =1.424 m2/ml |
|
50% is 5.85 microns |
Standard Deviation = 8.04 microns |
|
80% is 15.21 microns |
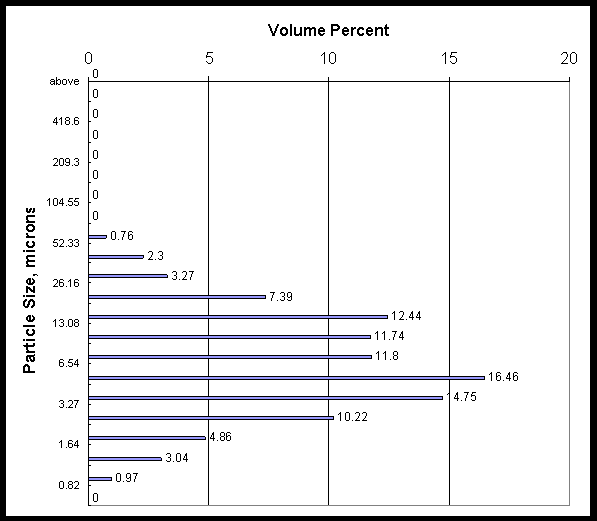
Data are the average of three measurements.
|
Mean value = 8.6 microns |
Mean Area = 4.19 microns |
|
20% is 2.82 microns |
Calculated Specific Area =1.434 m2/ml |
|
50% is 5.47 microns |
Standard Deviation = 6.11 microns |
|
80% is 13.26 microns |
Appendix C: Analysis of Filtrate Products
The following table provides the average analysis from three filtrates of the small C precipitation and filtration.
Analysis of Filtrate Products
|
Filt-A |
Filt-B |
Filt-Product |
Units |
|
|
Al |
7092.09 |
6977.338 |
7334.76 |
mg/liter |
|
B |
16.44 |
18.8 |
21.51 |
mg/liter |
|
Ba |
0.6 |
0.94 |
0.6 |
mg/liter |
|
Ca |
144.87 |
157.685 |
160.35 |
mg/liter |
|
Cd |
27.9 |
28.435 |
30.12 |
mg/liter |
|
Co |
1.98 |
2.35 |
2.16 |
mg/liter |
|
Cr |
105.66 |
107.019 |
86.07 |
mg/liter |
|
Cu |
26.49 |
26.508 |
23.37 |
mg/liter |
|
Fe |
3.96 |
5.405 |
5.31 |
mg/liter |
|
La |
3.9 |
6.11 |
2.4 |
mg/liter |
|
Li |
0.9 |
1.41 |
0.9 |
mg/liter |
|
Mg |
0.3 |
0.47 |
1.26 |
mg/liter |
|
Mn |
0.48 |
0.517 |
1.89 |
mg/liter |
|
Mo |
30.15 |
31.584 |
30.72 |
mg/liter |
|
Na |
126189.33 |
126240.543 |
137220 |
mg/liter |
|
Ni |
196.23 |
190.632 |
203.16 |
mg/liter |
|
P |
905.58 |
910.39 |
1324.77 |
mg/liter |
|
Pb |
60.12 |
61.899 |
69.81 |
mg/liter |
|
Si |
16.92 |
32.618 |
75.06 |
mg/liter |
|
Sn |
26.82 |
29.892 |
24.21 |
mg/liter |
|
Sr |
172.38 |
149.836 |
109.32 |
mg/liter |
|
Ti |
0.9 |
1.41 |
0.6 |
mg/liter |
|
V |
1.2 |
1.88 |
1.2 |
mg/liter |
|
Zn |
0.9 |
1.41 |
9 |
mg/liter |
|
Zr |
17.16 |
15.886 |
23.4 |
mg/liter |
|
Sr-90 |
3.94E+06 |
3.40E+06 |
1.64E+06 |
dpm/ml |
|
CS-137 |
4.09E+08 |
3.97E+08 |
4.38E+08 |
dpm/ml |
|
Am-241 |
3.30E+04 |
2.31E+04 |
2.99E+04 |
dpm/ml |
|
Cm-244 |
2.92E+04 |
1.92E+04 |
1.03E+04 |
dpm/ml |
|
Pu-239/240 |
2.85E+03 |
2.93E+03 |
4.13E+03 |
dpm/ml |
|
Pu-238 |
2.28E+03 |
1.69E+03 |
2.57E+03 |
dpm/ml |
|
Co-60 |
8.25E+04 |
9.00E+04 |
dpm/ml |
|
|
Cl |
0.046 |
0.029 |
0.041 |
molar |
|
F |
0.041 |
0.025 |
0.041 |
molar |
|
Formate |
0.100 |
0.062 |
0.113 |
molar |
|
Nitrate |
1.843 |
1.052 |
1.937 |
molar |
|
Nitrite |
0.811 |
0.479 |
0.853 |
molar |
|
Oxalate |
<0.034 |
<0.034 |
<0.034 |
molar |
|
Sulfate |
0.061 |
0.037 |
0.063 |
molar |
|
TIC |
3.54 |
8.52 |
10.9 |
g/liter |
|
TOC |
7.8 |
6 |
46.6 |
g/liter |
|
U Chemch |
7.8 |
4.8 |
7.8 |
mg/liter |
Appendix D: Composition of Filter Rig Flushes
The following tables provide compositions of the acid and final water flushes of the filter rig. The first table provides compositions for the flush acid and the final water flush after entrained solids filtration. The second table gives the same type of data for the last two flushes done after precipitate filtration.
Nitrate is relatvely high relative to the waste composition because nitric acid was used in the acid. Manganese and strontium are much higher in the second set of solutions compared to the first because of these added reagents in the precipitation.
Last Two Flush Solutions after Entrained Solids Filtration
|
Acid Flush |
error |
Water Flush |
error |
|
|
mg/liter |
% |
mg/liter |
% |
|
|
Al |
388.7 |
0.1 |
152.5 |
0.2 |
|
B |
0.9 |
0.2 |
||
|
Ba |
0.3 |
1.2 |
0.2 |
4.1 |
|
Ca |
12.8 |
3.7 |
6.6 |
10.0 |
|
Cd |
1.6 |
3.3 |
0.7 |
1.1 |
|
Co |
0.1 |
21.4 |
0.1 |
20.7 |
|
Cr |
10.7 |
1.3 |
6.4 |
2.6 |
|
Cu |
2.3 |
0.9 |
63.7 |
0.5 |
|
Fe |
8.6 |
2.0 |
23.2 |
2.1 |
|
La |
0.6 |
0.3 |
0.3 |
25.9 |
|
Li |
<0.03 |
0.1 |
21.4 |
|
|
Mg |
0.2 |
1.2 |
1.1 |
5.4 |
|
Mn |
2.5 |
0.7 |
1.9 |
1.3 |
|
Mo |
1.5 |
1.3 |
0.6 |
0.9 |
|
Na |
5865.8 |
3.9 |
2342.7 |
1.1 |
|
Ni |
10.9 |
3.1 |
7.5 |
6.1 |
|
P |
46.9 |
1.6 |
17.2 |
3.2 |
|
Pb |
5.8 |
5.6 |
6.4 |
5.7 |
|
Si |
1.6 |
5.7 |
1.3 |
17.8 |
|
Sn |
0.5 |
2.7 |
0.2 |
28.6 |
|
Sr |
0.1 |
0.9 |
0.1 |
5.3 |
|
Ti |
<0.03 |
0.1 |
29.7 |
|
|
V |
0.1 |
11.8 |
||
|
Zn |
1.2 |
1.0 |
2.7 |
1.3 |
|
Zr |
0.4 |
11.9 |
0.2 |
13.0 |
|
units |
dpm/ml |
% |
dpm/ml |
% |
|
Sr-90 |
4.82E+06 |
0.2 |
2.14E+06 |
11.2 |
|
CS-137 |
2.40E+07 |
2.4 |
8.96E+06 |
1.7 |
|
Am-241 |
16300 |
1.2 |
6545 |
2.9 |
|
Cm-244 |
7475 |
3.3 |
3475 |
3.2 |
|
Pu-239/240 |
641.5 |
17.0 |
427 |
55.3 |
|
Pu-238 |
428 |
6.1 |
202.5 |
2.5 |
|
Units |
Molar |
% |
Molar |
% |
|
Cl |
0.00206 |
0.0 |
0.000945 |
1.5 |
|
F |
0.00266 |
1.0 |
0.001 |
5.3 |
|
Formate |
0.00471 |
5.7 |
0.00168 |
3.3 |
|
Nitrate |
0.662 |
0.0 |
0.198 |
0.9 |
|
Nitrite |
not det. |
not det. |
||
|
Oxalate |
not det. |
not det. |
||
|
Phosphate |
not det. |
not det. |
||
|
Sulfate |
0.003589 |
1 |
0.001391 |
2.6 |
|
g/liter |
% |
g/liter |
% |
|
|
TIC |
not det. |
not det. |
||
|
TOC |
0.3985 |
-0.4 |
0.311 |
2.3 |
|
mg/liter |
% |
mg/liter |
% |
|
|
U Chemch |
0.715 |
2.1 |
0.245 |
6.1 |
Last Two Flush Solutions after Precipitation Filtration
|
precipitate washes |
|||
|
Element |
Acid Flush |
Water Flush |
|
|
mg/liter |
mg/liter |
||
|
Al |
475.062 |
63.731 |
|
|
B |
1.453 |
0.294 |
|
|
Ba |
0.561 |
0.146 |
|
|
Ca |
14.717 |
2.77 |
|
|
Cd |
2.047 |
0.424 |
|
|
Co |
0.281 |
0.176 |
|
|
Cr |
10.397 |
1.68 |
|
|
Cu |
1.733 |
0.38 |
|
|
Fe |
9.079 |
1.626 |
|
|
La |
0.551 |
0.244 |
|
|
Li |
0.046 |
0.066 |
|
|
Mg |
0.072 |
0.01 |
|
|
Mn |
137.005 |
17.729 |
|
|
Mo |
2.115 |
0.343 |
|
|
Na |
8753.099 |
1252.235 |
|
|
Ni |
15.017 |
2.696 |
|
|
P |
60.365 |
7.378 |
|
|
Pb |
7.290 |
1.464 |
|
|
Si |
7.582 |
1.924 |
|
|
Sn |
0.937 |
0.264 |
|
|
Sr |
304.365 |
42.132 |
|
|
Ti |
0.092 |
0.113 |
|
|
V |
0.141 |
0.17 |
|
|
Zn |
0.766 |
0.069 |
|
|
Zr |
0.357 |
0.202 |
|
|
Units |
dpm/ml |
dpm/ml |
|
|
Sr-90 |
3.95E+06 |
3.35E+06 |
|
|
CS-137 |
2.77E+07 |
4.02E+06 |
|
|
Am-241 |
1.13E+04 |
1.53E+03 |
|
|
Cm-244 |
6.48E+03 |
2.00E+03 |
|
|
Pu-239/240 |
202 |
267 |
|
|
Pu-238 |
66 |
65 |
|
|
units |
Molar |
Molar |
|
|
Cl |
0.002849 |
0.000508 |
|
|
F |
0.003105 |
0.000368 |
|
|
Formate |
0.007289 |
0.001022 |
|
|
Nitrate |
0.74379 |
0.059935 |
|
|
Nitrite |
0 |
0.001217 |
|
|
Oxalate |
0 |
0 |
|
|
Sulfate |
0.004552 |
0.000573 |
|
|
TIC |
9.3 |
0.048 |
g/l |
|
TOC |
not det. |
6.48 |
|
|
U Chemch |
0.62 |
2.1 |
mg/l |