
WSRC-TR-2000-00262
Recommended Partition Coefficient (Kd)
Values for Nuclide Partitioning
in the Presence of Cellulose Degradation Products
S. M. Serkiz
Westinghouse Savannah River Company
Aiken, SC 20908
This report was prepared as an account of work sponsored by an agency of the United States Government. Neither the United States Government nor any agency thereof, nor any of their employees, makes any warranty, express or implied, or assumes any legal liability or responsibility for the accuracy, completeness, or usefulness of any information, apparatus, product or process disclosed, or represents that its use would not infringe privately owned rights. Reference herein to any specific commercial product, process or service by trade name, trademark, manufacturer, or otherwise does not necessarily constitute or imply its endorsement, recommendation, or favoring by the United States Government or any agency thereof. The views and opinions of authors expressed herein do not necessarily state or reflect those of the United States Government or any agency thereof.
This report has been reproduced directly from the best available copy.
Available for sale to the public, in paper, from: U.S. Department of Commerce, National Technical Information Service, 5285 Port Royal Road, Springfield, VA 22161, phone: (800) 553-6847, fax: (703) 605-6900, email: orders@ntis.fedworld.gov online ordering: http://www.ntis.gov/support/ordering.htm
Available electronically at http://www.osti.gov/bridge/
Available for a processing fee to U.S. Department
of Energy and its contractors, in paper, from: U.S. Department of Energy, Office
of Scientific and Technical Information, P.O. Box 62, Oak Ridge, TN 37831-0062,
phone: (865 ) 576-8401, fax: (865) 576-5728, email: reports@adonis.osti.gov
Executive Summary
Degradation products of cellulosic materials (e.g., paper and wood products) can significantly influence the subsurface transport of metals and radionuclides. Co-disposal of radionuclides with cellulosic materials in the E-Area slit trenches at the SRS is, therefore, expected to influence nuclide fate and transport in the subsurface.
Initial modeling of this system was undertaken in 1996 and utilized published stability constants for reactions between low molecular weight acids (i.e., citric acid and EDTA) and numerous radionuclides. In this approach, the physicochemical properties of these simple organic acids were intended to approximate effects of cellulose degradation products (CDP) on nuclide sorption. Notable differences in the allowable radionuclide inventory of the E-Area Slit Trench were predicted when the presence of CDP was accounted for in this conceptual model (Serkiz and Myers, 1996).
A series of laboratory studies were initiated in April 1998 to validate and/or update the modeling assumptions employed in the 1996 modeling effort. In these studies, laboratory results were generated for ternary systems containing divalent and trivalent radionuclides (uranium (VI) or europium (III)), organic matter (Suwannee River dissolved organic matter or laboratory generated CDP), and a soil phase (SRS Burial Ground soil or Aiken kaolinite) (Serkiz et al., 1998 and Serkiz et al., 1999). Laboratory data on the influence of CDP on metal/nuclide cation sorption show that these organic compounds can significantly reduce sorption, both directly and indirectly. Directly through complexation of metals in the aqueous phase, thus, inhibiting sorption and indirectly by reducing the system pH that, in turn, results in a more positively charged soil surface also inhibiting cation sorption. The direct effects of organic matter at more neutral pH values is several orders of magnitude reduction under conditions of high organic matter content. At low pH values, Kd values are similar regardless of organic matter content. Kd variability due to pH, at constant organic carbon content, is as high as about two orders of magnitude. The degree of this pH effect decreases with increasing organic matter content. Furthermore, these studies showed that dissolved organic matter production from cellulosic source materials was a very dynamic process with concentrations over 1 g per liter of organic carbon and pH values below 4.5 observed in laboratory static leaching tests.
Keywords: Radionuclide Speciation and Transport, Cellulose
Degradation, Dissolved Organic Matter,
Europium, Uranium, E-Area Vaults, Performance Assessment
Introduction
This report documents the data analysis of the results of the above described laboratory studies in order to recommend Kd values for use in Performance Assessment modeling of nuclide transport in the presence of CDP.
Because generating laboratory data on the influence of CDP on the mobility of the 29 elements included in the E-Area Vaults PA modeling effort would be cost and time prohibitive, an indexing approach based on chemical analogues was adopted for cationic species. The index approach taken in this work is largely empirical and should be validated with additional studies both in the field and laboratory.
The highest DOC concentrations are expected in the vicinity of the actual waste. These concentrations, and the resulting effect on Kd values, will decrease with distance from the waste. This effect should be investigated, both in the field and with modeling studies.
Methods
In recommending CDP influenced Kd values for use in PA modeling, both the primary (interactions of CDP with both the soil surface and the dissolved metal/nuclide) and secondary (reactions involving CDP that impact major-ion chemistry (e.g., pH)) influences of the CDP on metal/nuclide sorption were considered. The general conceptual model is:
The general approach to generating CDP-influenced Kd values was to:
Curve Fitting
This section describes the curve fitting activities employed on laboratory data for the ternary system of organic matter/SRS soil material/nuclide.
Laboratory data for the sorption of divalent uranyl (UO22+) and trivalent europium (Eu3+) to SRS soils or soil components in the presence of both naturally occurring dissolved organic matter and laboratory generated CDP formed the basis for evaluating the direct affect of CDP on divalent/trivalent cation sorption. In order to facilitate Kd calculations, a three-parameter logistic function was fitted to the experimental data in the form of pH versus fraction metal/radionuclide sorbed. To minimize model-fitting biases, data sets were truncated to pH values of less than 6.0 prior to model fitting. The mathematical form of the three-parameter logistic function is:

Where: FS is the fraction metal/nuclide sorbed to the soil; a is the y asymptote and is a fitting parameter with a limit set to less than 1; pH0 is the pH at the inflection point of the curve and is a fitting parameter; and b is a fitting parameter that relates to the steepness of the rise of the curve. The general shape of the curve is a sigmoid whose fraction of metal/nuclide sorbed ranges between zero and one and, for the cation sorption data analyzed, starts low at low-pH values and increases at higher values.
Kd is related to the FS by:

Where S/L is the solid to liquid ratio under which the data were generated.
Results
This section describes the results of the general approach to estimate CDP-influenced Kd values described above.
Organic Carbon Content/pH Relationship
In a previous laboratory study, actual cellulose degradation products were generated in the laboratory from materials known to be present in slit trenches at SRS (Serkiz et al., 1999). As expected, the pH of the cellulose leachate slowly decreased with time (Figure 1) from 5.2 at the start to 4.4 after 19 weeks. This is consistent with the production of carboxylic acid and hydroxyl functional groups from wood degradation processes. After an initial large production of dissolved organic carbon (DOC) in the first week, the DOC of the samples steadily increased from 545 mg C/L at week 1, to 1018 mg C/L at week 19 (Figure 1). The slope of the total organic carbon (TOC) data as a function of time (i.e., organic carbon production rate) decreases as the experiment progresses.
Based on these experimental data and soil-pH for the soil used in the laboratory sorption study (Johnson, 1995), a relationship between organic carbon content and pH was developed to facilitate further Kd calculations. This relationship is summarized in Table 1.

Curve Fitting
Laboratory data for the ternary system containing the trivalent lanthanide europium, organic matter (Suwannee River DOM and CDP), and SRS soil are contained in Serkiz et al. (1998) and Serkiz et al. (1999). Data at 0, 10, 30, 100, and 500 mg C/L were fitted to the three-parameter logistic function as described above. The curve fitting data are summarized in Tables 2 through 6 for 0, 10, 30, 100, and 500 mg C/L laboratory data respectively. Graphical representations of the laboratory data and model fits are shown in Figures 2 through 6 for the 0, 10, 30, 100, and 500 mg C/L laboratory data respectively.
Table 2 – Curve Fitting Summary Eu Sorption to SRS Soil No Added
Organic Matter
(Fitted to Data in Figure 2)
|
Eu Sorption Experiments |
3 Parameter Logistic Fit |
|||
|
[DOM] = |
0 mgC/L |
a = |
0.995 |
|
|
Solid |
SRS Soil |
b = |
-12.107 |
|
|
S/L (g/mL) |
0.0238 |
pH0 = |
2.810 |
|
|
Model Predictions |
||||
|
pH |
Fraction Sorbed |
Kd |
||
|
4.50 |
0.9913 |
4765 |
||
|
4.75 |
0.9928 |
5832 |
||
|
5.00 |
0.9936 |
6568 |
||
|
5.25 |
0.9941 |
7028 |
||
|
5.50 |
0.9943 |
7301 |
||
|
5.75 |
0.9944 |
7460 |
||
|
6.00 |
0.9945 |
7554 |
||
Table 3 – Curve Fitting Summary Eu Sorption to SRS Soil 10 mg C/L Suwannee River
Organic Matter (Fitted to Data in Figure 3)
|
Eu Sorption Experiments |
3 Parameter Logistic Fit |
|||
|
[DOM] = |
10 mgC/L |
a = |
1.000 |
|
|
Solid |
SRS Soil |
b = |
-7.247 |
|
|
S/L (g/mL) |
0.0238 |
pH0 = |
3.279 |
|
|
Model Predictions |
||||
|
pH |
Fraction Sorbed |
Kd |
||
|
4.50 |
0.9084 |
416 |
||
|
4.75 |
0.9362 |
616 |
||
|
5.00 |
0.9551 |
894 |
||
|
5.25 |
0.9681 |
1273 |
||
|
5.50 |
0.9770 |
1783 |
||
|
5.75 |
0.9832 |
2460 |
||
|
6.00 |
0.9876 |
3349 |
||
Table 4 – Curve Fitting Summary Eu Sorption to SRS Soil 30 mg C/L Suwannee River
Organic Matter (Fitted to Data in Figure 4)
|
Eu Sorption Experiments |
3 Parameter Logistic Fit |
|||
|
[DOM] = |
30 mgC/L |
a = |
0.835 |
|
|
Solid |
SRS Soil |
b = |
-11.992 |
|
|
S/L (g/mL) |
0.0238 |
pH0 = |
3.090 |
|
|
Model Predictions |
||||
|
pH |
Fraction Sorbed |
Kd |
||
|
4.50 |
0.8260 |
199 |
||
|
4.75 |
0.8303 |
206 |
||
|
5.00 |
0.8325 |
209 |
||
|
5.25 |
0.8337 |
211 |
||
|
5.50 |
0.8343 |
211 |
||
|
5.75 |
0.8346 |
212 |
||
|
6.00 |
0.8348 |
212 |
||
Table 5 – Curve Fitting Summary Eu Sorption to SRS Soil 100 mg C/L Suwannee River
Organic Matter (Fitted to Data in Figure 5)
|
Eu Sorption Experiments |
3 Parameter Logistic Fit |
|||
|
[DOM] = |
100 mgC/L |
a = |
0.702 |
|
|
Solid |
SRS Soil |
b = |
5.346 |
|
|
S/L (g/mL) |
0.0238 |
pH0 = |
7.084 |
|
|
Model Predictions |
||||
|
pH |
Fraction Sorbed |
Kd |
||
|
4.50 |
0.6447 |
76 |
||
|
4.75 |
0.6277 |
71 |
||
|
5.00 |
0.6074 |
65 |
||
|
5.25 |
0.5840 |
59 |
||
|
5.50 |
0.5576 |
53 |
||
|
5.75 |
0.5285 |
47 |
||
|
6.00 |
0.4971 |
42 |
||
Table 6 – Curve Fitting Summary Eu Sorption to SRS Soil 500 mg C/L Suwannee River
Organic Matter (Fitted to Data in Figure 6)
|
Eu Sorption Experiments |
3 Parameter Logistic Fit |
|||
|
[DOM] = |
500 mgC/L |
a = |
0.581 |
|
|
Solid |
SRS Soil |
b = |
2.004 |
|
|
S/L (g/mL) |
0.0238 |
pH0 = |
11.353 |
|
|
Model Predictions |
||||
|
pH |
Fraction Sorbed |
Kd |
||
|
4.50 |
0.5024 |
42 |
||
|
4.75 |
0.4947 |
41 |
||
|
5.00 |
0.4869 |
40 |
||
|
5.25 |
0.4789 |
39 |
||
|
5.50 |
0.4708 |
37 |
||
|
5.75 |
0.4626 |
36 |
||
|
6.00 |
0.4544 |
35 |
||
Laboratory data for the ternary system containing the divalent uranyl ion, organic matter (Suwannee River DOM), and SRS soil components (kaolinite and Burial Ground soil) are contained in Serkiz et al. (1998). Uranyl sorption data to kaolinite at 0, 10, and 30 mg C/L and uranyl sorption to an SRS Burial Ground soil in the absence of added organic matter (Johnson, 1995) were fitted to the three parameter logistic function as described above. The curve fitting data are summarized in Tables 7 through 9 for 0, 10, and 30 mg C/L sorption to kaolinite laboratory data respectively and Table 10 for uranyl sorption to SRS soil in the absence of added organic matter. Graphical representations of the laboratory data and model fits are shown in Figures 7 through 9 for the 0, 10, and 30 mg C/L laboratory data respectively and Figure 10 for uranyl sorption to SRS soil in the absence of added organic matter. The kaolinite Kd data were adjusted to a whole soil response by assuming a clay content of 3.8% by weight and that the remaining soil mass was unreactive with respect to to uranyl sorption.
Table 7 – Curve Fitting Summary U Sorption to Kaolinite with
0 Added
Organic Matter (Fitted to Data in Figure 7)
|
U Sorption Data From Turner (1995) |
3 Parameter Logistic Fit |
|||
|
[DOM] = |
0 mgC/L |
a = |
1.000 |
|
|
Solid |
Kaolinite |
b = |
-21.049 |
|
|
S/L Kaolinite |
0.004 |
pH0 = |
4.715 |
|
|
S/L (g/mL) |
0.105 |
|||
|
Model Predictions |
||||
|
pH |
Fraction Sorbed |
Kd |
||
|
4.50 |
0.2720 |
4 |
||
|
4.75 |
0.5384 |
11 |
||
|
5.00 |
0.7744 |
33 |
||
|
5.25 |
0.9056 |
91 |
||
|
5.50 |
0.9623 |
242 |
||
|
5.75 |
0.9849 |
618 |
||
|
6.00 |
0.9938 |
1514 |
||
Table 8 – Curve Fitting Summary U Sorption to Kaolinite 10 mg C/L Suwannee River
Organic Matter (Fitted to Data in Figure 8)
|
U Sorption Data From Serkiz et al., 1998 |
3 Parameter Logistic Fit |
|||
|
[DOM] = |
10 mgC/L |
a = |
0.991 |
|
|
Solid |
Kaolinite |
b = |
-9.362 |
|
|
S/L Kaolinite |
0.003 |
pH0 = |
3.606 |
|
|
S/L (g/mL) |
0.0789 |
|||
|
Model Predictions |
||||
|
pH |
Fraction Sorbed |
Kd |
||
|
4.50 |
0.8805 |
93 |
||
|
4.75 |
0.9214 |
148 |
||
|
5.00 |
0.9468 |
225 |
||
|
5.25 |
0.9626 |
326 |
||
|
5.50 |
0.9725 |
448 |
||
|
5.75 |
0.9788 |
584 |
||
|
6.00 |
0.9828 |
724 |
||
Table 9 – Curve Fitting Summary U Sorption to Kaolinite 30 mg C/L Suwannee River
Organic Matter (Fitted to Data in Figure 9)
|
U Sorption Data From Serkiz et al., 1998 |
3 Parameter Logistic Fit |
|||
|
[DOM] = |
30 mgC/L |
a = |
0.562 |
|
|
Solid |
Kaolinite |
b = |
-30.459 |
|
|
S/L Kaolinite |
0.003 |
pH0 = |
3.180 |
|
|
S/L (g/mL) |
0.0789 |
|||
|
Model Predictions |
||||
|
pH |
Fraction Sorbed |
Kd |
||
|
4.50 |
0.5621 |
16 |
||
|
4.75 |
0.5621 |
16 |
||
|
5.00 |
0.5621 |
16 |
||
|
5.25 |
0.5621 |
16 |
||
|
5.50 |
0.5621 |
16 |
||
|
5.75 |
0.5621 |
16 |
||
|
6.00 |
0.5621 |
16 |
||
Table 10 – Curve Fitting Summary U Sorption to SRS Soil 0 Added Organic Matter
(Fitted to Data in Figure 10)
|
U Sorption Data From Johnson (1995) |
3 Parameter Logistic Fit |
|||
|
[DOM] = |
0 mgC/L |
a = |
1.000 |
|
|
Solid |
SRS Soil |
b = |
-14.660 |
|
|
S/L (g/mL) |
0.0238 |
pH0 = |
4.498 |
|
|
Model Predictions |
||||
|
pH |
Fraction Sorbed |
Kd |
||
|
4.50 |
0.5014 |
42 |
||
|
4.75 |
0.6896 |
93 |
||
|
5.00 |
0.8249 |
198 |
||
|
5.25 |
0.9060 |
405 |
||
|
5.50 |
0.9501 |
800 |
||
|
5.75 |
0.9734 |
1536 |
||
|
6.00 |
0.9856 |
2866 |
||
The variability in Kd with respect to changes in both pH and organic carbon content is clearly shown in the laboratory data. The variability due to pH, at a constant organic carbon content, is as high as about two orders of magnitude. The size of this pH effect decreases with increasing organic matter content. Similarly, the effects of organic matter at more neutral pH values is several orders of magnitude reduction at high organic matter content. At low-pH values, however, Kd values are similar regardless of organic matter content.
Recommended Kd values, along with the fractional decrease in Kd with increasing organic matter content, based on laboratory data for ternary systems of Eu or U, natural organic matter, and SRS soil components are summarized in Table 11.
Table 11 – Summary of Eu and U Kd Values From Laboratory Data
|
Model Regime DOC Range (mgC/L) |
DOC Data Used in Calc. (mgC/L) |
pH |
Trivalent Eu Kd (mL/g) |
Fraction of Eu-Kd with [DOC]=0 |
Divalent UO2 Kd (mL/g) |
Fraction of UO2Kd with [DOC]=0 |
|
< 1 |
0 |
5.50 |
7300 |
1.00 |
800 |
1.00 |
|
10 to 1 |
10 |
5.25 |
1300 |
0.18 |
325 |
0.41 |
|
30 to 10 |
30 |
5.00 |
210 |
0.029 |
16 |
0.20 |
|
100 to 30 |
100 |
4.75 |
70 |
0.0096 |
8 |
0.0096 |
|
1000 to 100 |
500 |
4.50 |
40 |
0.0055 |
4 |
0.0055 |

Site-specific data for the sorption of Cs to SRS soils as a function of pH are from work presented in Johnson (1995). These data are summarized in Table 12 for the pH in each of the model regimes. As expected, the fractional Kd reduction with decreasing pH is much less than for the multivalent cations. Because organic matter is not expected to strongly influence monovalent cation sorption directly, only the effect of pH on monovalent cation sorption was considered. Similar to the approach taken with the multivalent cation species, other monocations were indexed to the Kd reductions observed with decreasing pH for Cs sorption to SRS soil.
Table 12 – Summary of Cs Kd Values From Laboratory Data
|
Model Regime DOC Range (mgC/L) |
DOC Data Used in Calc. (mgC/L) |
pH |
Cs Kd (mL/g) |
Fraction of Baseline Kd |
|
< 1 |
0 |
5.50 |
18 |
1.00 |
|
10 to 1 |
0 |
5.25 |
12 |
0.67 |
|
30 to 10 |
0 |
5.00 |
5 |
0.28 |
|
100 to 30 |
0 |
4.75 |
5 |
0.28 |
|
1000 to 100 |
0 |
4.50 |
5 |
0.28 |
Based on the approach described above, recommended CDP-influenced Kd values for the 29 elements considered in E-Area Vaults Disposal Facility were calculated. Where site-specific Kd data were available (i.e., U, Cd, Cs, and Eu) for nuclide sorption in the absence of organic matter, they were recommended for use in the less than one mgC/L model regime and in calculating Kd values for all other model regimes. The results from this exercise and summary information from the previous PA modeling effort (McDowell-Boyer, 2000) are presented in Table 13.
|
Table 13 - Recommended Kd Values |
||||||||
|
--------------[DOC(mgC/L]----------- |
||||||||
|
--E-Area Vault PA Data (2000)--- |
1 |
10 |
30 |
100 |
1000 |
|||
|
Nuclides GW |
Kd (ml/g) |
5.5 |
5.25 |
5.00 |
4.75 |
4.5 |
Notes |
|
|
Ac |
Ac+3 |
450 |
450 |
80 |
13 |
4 |
2 |
Scaled From Knaub's Eu Data and PA Kd |
|
Am |
Am3+ |
1900 |
1900 |
338 |
55 |
18 |
10 |
Scaled From Knaub's Eu Data and PA Kd |
|
C |
CO3-2 |
2 |
2 |
2 |
2 |
2 |
2 |
Use PA Kd (Form of C not Known) |
|
Cf |
Cf3+ |
510 |
510 |
91 |
15 |
5 |
3 |
Scaled From Knaub's Eu Data and PA Kd |
|
Cm |
Cm3+ |
4000 |
4000 |
712 |
115 |
38 |
22 |
Scaled From Knaub's Eu Data and PA Kd |
|
Cs |
Cs+ |
330 |
18 |
12 |
5 |
5 |
5 |
Johnson's SRS Data 0 DOM |
|
Eu |
Eu+3 |
NA |
7300 |
1300 |
210.0 |
70.0 |
40.0 |
From Dave Knaub's Data 0,10,30,100, 500 mgC/l |
|
H |
HTO |
0 |
0 |
0 |
0 |
0 |
0 |
Kd of 0 used in PA |
|
I |
I- |
0.6 |
0.6 |
0.6 |
0.6 |
0.6 |
0.6 |
Anionic Assume No Effect of pH or DOM |
|
Nb |
Anionic |
160 |
160 |
160 |
160 |
160 |
160 |
Anionic Assume No Effect of pH or DOM |
|
Ni |
Ni+2 |
400 |
400 |
163 |
8 |
3.8 |
2.2 |
Scaled From Uranium Kd Response |
|
Np |
NpO2+1 |
5 |
5 |
3 |
1 |
1 |
1 |
Scaled From Johnson (1995) Data and PA Kd |
|
Pa |
PaO2+1 |
550 |
550 |
3 |
1.4 |
1.4 |
1.4 |
Scaled From Johnson (1995) Data and PA Kd |
|
Pb |
Pb+2 |
270 |
270 |
110 |
5 |
3 |
1 |
Scaled From Uranium Kd Response |
|
Pd |
Pd+2 |
55 |
55 |
22.34 |
1.1 |
0.527 |
0.301 |
Scaled From Uranium Kd Response |
|
Po |
Po-2 |
150 |
150 |
150 |
150 |
150 |
150 |
Anionic Assume No Effect of pH or DOM |
|
Pu |
Pu4+ |
100 |
100 |
18 |
3 |
1 |
1 |
Scaled From Knaub's Eu Data and PA Kd |
|
Ra |
Ra+2 |
500 |
500 |
203.1 |
10 |
4.795 |
2.74 |
Scaled From Uranium Kd Response |
|
Rb |
Rb+ |
55 |
55 |
3 |
1 |
1 |
1 |
Scaled From Johnson (1995) Data and PA Kd |
|
Se |
SeO3-2 |
5 |
5 |
5 |
5 |
5 |
5 |
Anionic Assume No Effect of pH or DOM |
|
Sn |
Sn+2 |
130 |
130 |
52.81 |
2.6 |
1.247 |
0.712 |
Scaled From Uranium Kd Response |
|
Sr |
Sr2+ |
10 |
10 |
4 |
0.2 |
0.1 |
0.1 |
Scaled From Uranium Kd Response |
|
Tc |
TcO4- |
0.36 |
0.36 |
0.36 |
0.36 |
0.36 |
0.36 |
Anionic Assume No Effect of pH or DOM |
|
Th |
Th+4 |
3200 |
3200 |
570 |
92 |
31 |
18 |
Scaled From Knaub's Eu Data and PA Kd |
|
U |
UO2+2 |
35 |
800 |
325 |
16 |
8 |
4 |
1,10, 30 mgC/L for Lab Data (Johnson and Uhal) 100, 1000 scaled from Eu Data |
|
Zr |
Zr+4 |
600 |
600 |
107 |
17 |
6 |
3 |
Scaled From Knaub's Eu Data and PA Kd |
|
Notes: |
recommended CDP unimpacted Kd different than PA data |
|||||||
|
NA |
Not Available |
|||||||
Conclusions
Laboratory data on the influence of CDP on metal/nuclide cation sorption show that these organic compounds can significantly reduce sorption, both directly and indirectly. Directly through complexation of metals in the aqueous phase, thus, inhibiting sorption and indirectly by reducing the system pH that, in turn, results in a more positively charged soil surface also inhibiting cation sorption. The direct effects of organic matter at more neutral pH values is several orders of magnitude reduction under conditions of high organic matter content. At low pH values, Kd values are similar regardless of organic matter content. Kd variability due to pH, at constant organic carbon content, is as high as about two orders of magnitude. The degree of this pH effect decreases with increasing organic matter content.
Because generating laboratory data on the influence of CDP on the mobility of the 29 elements included in the E-Area Vaults PA modeling effort would be cost and time prohibitive, an indexing approach based on chemical analogues was adopted for cationic species. As expected, the higher the cationic charge of the specie of interest the greater the influence of the organic matter, over the geochemical range found in the model regimes. Using this indexing approach, Kd reductions of over 99 percent were calculated. Given the large Kd reduction for this index approach, if a high degree of conservatism is incorporated in the PA Kd value, then unrealistically low inventories for certain nuclides could be calculated.
The index approach taken in this work is largely empirical and should be validated with additional studies both in the field and laboratory. Certain mechanisms not accounted for in this work could significantly increase the allowable PA inventory if they are incorporated in the PA models. For example metal/nuclide reduction could result in a very high Kd for U, Pu, and, Tc by the virtue of a low solubility limit on the reduced form of these elements. The highest DOC concentrations are expected in the vicinity of the actual waste. These concentrations, and the resulting effect on Kd values, will decrease with distance from the waste. This effect should be investigated, both in the field and with modeling studies. Although the pH buffering capacities of SRS soils are relatively low, this was not accounted for in estimating the magnitude of the pH reduction due to the formation of CDP. Some credit for this soil-buffering mechanism could be accounted for with additional laboratory studies.
References
Figures
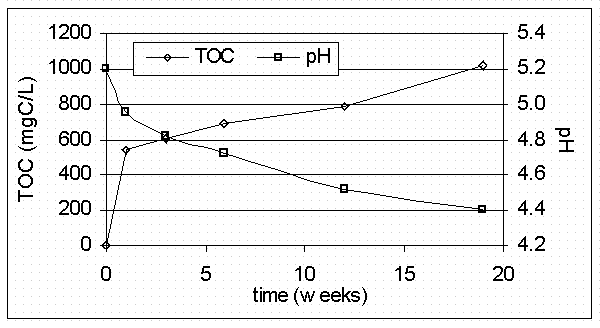
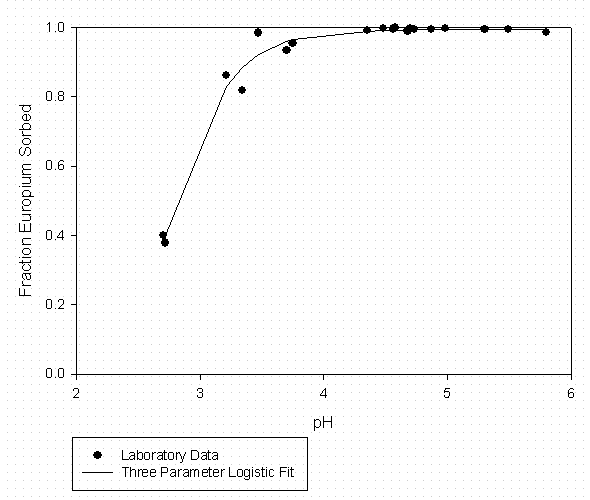
Figure 2 – Eu Sorption to SRS Soil (24 g/L) Suwannee River DOM at 0 mgC/l
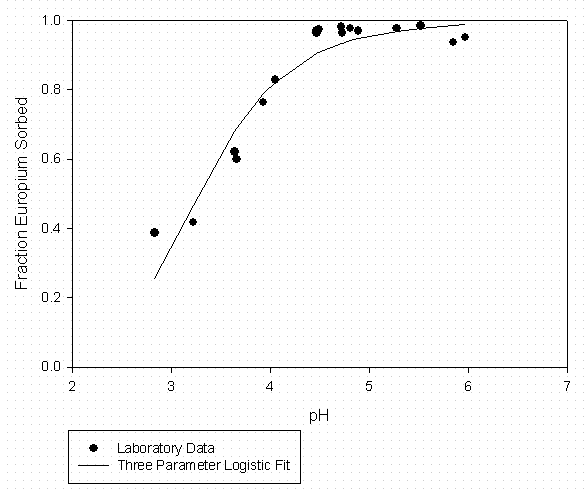
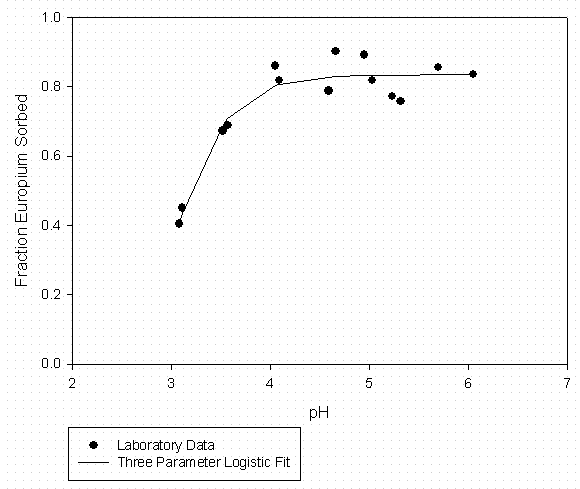
Figure 4 - Eu Sorption to SRS Soil (24 g/L) Suwannee River DOM at 30 mgC/l
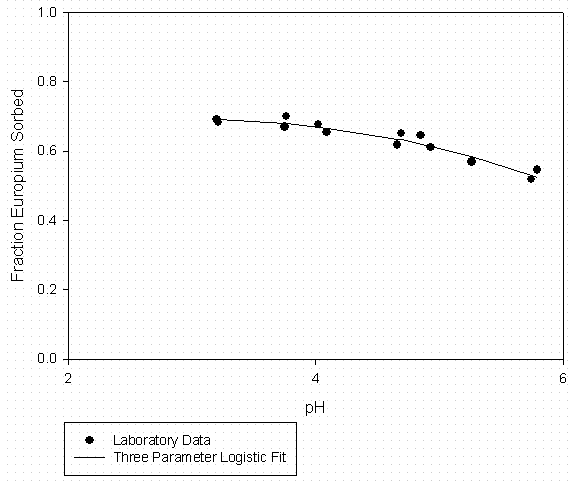
Figure 5 - Eu Sorption to SRS Soil (24 g/L) Suwannee River DOM at 100 mgC/l
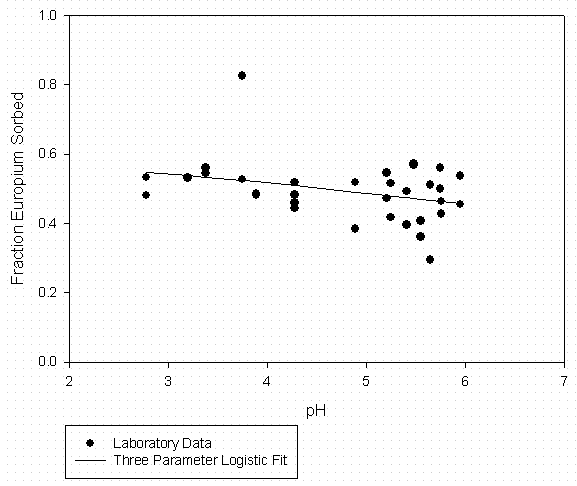
Figure 6 - Eu Sorption to SRS Soil (24 g/L) Suwannee River DOM at 500 mgC/l
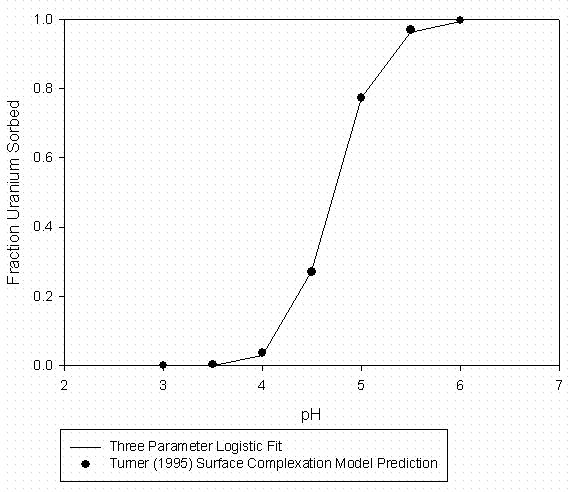
Figure 7 - U Sorption to Kaolinite (4 g/L) Data from Turner (1995) No Added Organic Matter
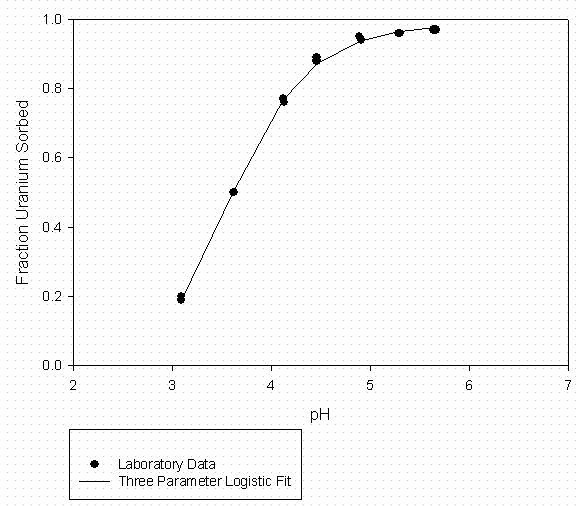
Figure 8 - U Sorption to Aiken Kaolinite (3g/L) Suwannee River DOM at 10 mgC/l
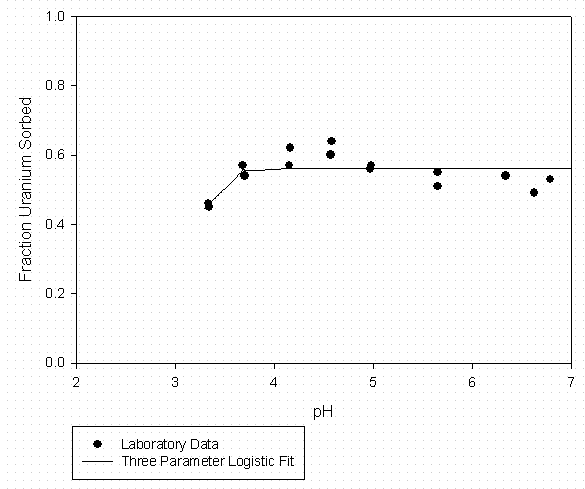
Figure 9 -U Sorption to Aiken Kaolinite (3g/L) Suwannee River DOM at 30 mgC/l
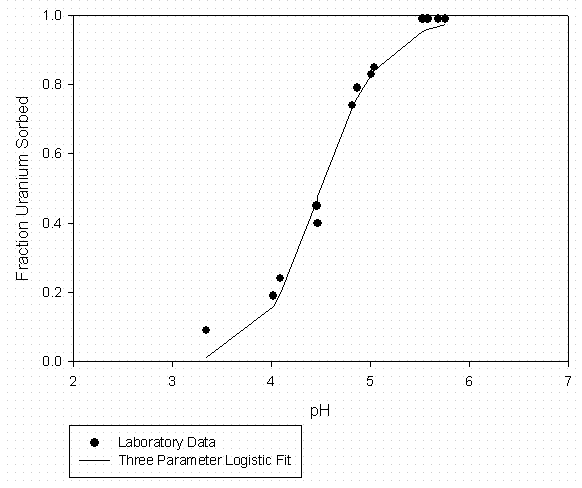
Figure 10 - U Sorption to SRS Soil (2.38 g/L) From Johnson (1995) No Added Organic Matter