This report was prepared as an account of work sponsored by an agency of the United States Government. Neither the United States Government nor any agency thereof, nor any of their employees, makes any warranty, express or implied, or assumes any legal liability or responsibility for the accuracy, completeness, or usefulness of any information, apparatus, product or process disclosed, or represents that its use would not infringe privately owned rights. Reference herein to any specific commercial product, process or service by trade name, trademark, manufacturer, or otherwise does not necessarily constitute or imply its endorsement, recommendation, or favoring by the United States Government or any agency thereof. The views and opinions of authors expressed herein do not necessarily state or reflect those of the United States Government or any agency thereof.
This report has been reproduced directly from the best available copy.
Available to DOE and DOE Contractors from the Office of Scientific and
Technical Information, P. O. Box 62 Oak Ridge, TN 37831; prices available from
(423) 576-8401. Available to the public from the National Technical Information
Service, U.S. Department of Commerce, 5285 Port Royal Road, Springfield, VA
22161.
A correction is needed to calculate the concentration of airborne tritium oxide when dried silica gel is used as the collector. This tracer study with tritiated water shows that the concentration of tritium in the water desorbed from silica gel is lower than in the adsorbed water by a fraction that increases with the amount of adsorbed water. The hypothesis was tested that the tritium in adsorbed tritiated water is diluted by isotopic exchange with non-tritiated water and hydroxyl groups in the silica gel collector. The extent of dilution was measured from 4% to 14% adsorbed water, which is typical of moisture on field collectors for monitoring airborne tritium oxide. For this entire range of percent adsorbed water, the inferred percent exchangeable water in the silica gel under study was 6.3 +/- 0.1%. This value compares to the silica gel weight loss of 5.3% by heating to 1,050oC. An explanation of the difference is proposed. The contribution of the HTO/H2O vapor pressure isotope effect was considered in calculating isotopic exchange. A curve is presented for correcting the measured tritium concentration in the distillate from the silica gel as a function of the amount of adsorbed water. The tritium tracer procedure is recommended for determining the percent exchangeable water in other silica gels to correct tritium measurements of water vapor collected by them.
Key words: silica gel, water adsorption, tritium monitoring, isotopic exchange, isotope effect
A common method for measuring tritium oxide in air is to collect it on silica gel from air that is pumped through a column at a measured flow rate. At the end of the collection period, the column is weighed to determine the amount of collected water, the water is distilled from the silica gel, and tritium in the distillate is measured with a liquid scintillation (LS) counter. One concern that apparently has been overlooked is that silica gels dried at 100oC still contain water and hydroxyl groups. If isotopic exchange occurs, the tritiated water that is measured will consist of adsorbed tritiated water vapor diluted with non-tritiated water from water and hydroxyl groups initially on the silica gel. This study examined the hypothesis of isotopic exchange of HTO with a constant fraction of H2O and -OH on silica gel by comparing tritium concentrations in water adsorbed on and desorbed from a column on which various amounts of water vapor had been collected.
Silica gel columns have been used at major nuclear facilities for environmental surveillance of airborne tritium oxide (Arnett and Mamatey 1997; Patton et al. 1997; Thompson et al. 1978; Bishop et al. 1985). Tests of silica gel columns by Patton et al. (1997) show the extent and pattern of tritiated water retention by silica gel but did not consider the cause of decreased tritium concentrations in desorbed water.
The complex interactions of silica gel with water have been the subject of numerous studies; one finding is that water and hydroxyl groups (i.e., silanol, =SiOH) that yield water upon heating are associated with silica gel at temperatures well above 100oC (Iler 1979; Unger 1979; Bergna 1994). The water and hydroxyl groups may be considered to be within the silica gel structure, in micropores, and on surfaces. Spectroscopic analysis is used to differentiate among types of bonds (Bergna 1994). A simple distinction among bonds is provided by the step-wise removal of water in several temperature ranges. Scott and Traiman (1980) reported three distinct groups (for a silica gel different from the one used here), one of which is removed by heating below 120o C., the second, below 650o C., and the third, above 750o C. Attempts to remove all intrinsic water at elevated temperature can destroy the structure of the silica gel.
In a study to measure the surface hydroxyl group concentration in silica gel with tritiated water vapor by isotope exchange, it was noted that an isotope effect also has to be considered (Unger and Gallei 1970; Unger 1979). The contribution of the HTO/H2O vapor pressure isotope effect can be calculated in terms of the isotope separation factor a, which can be defined for dilute tritiated water as the ratio of the HTO concentration in the condensed phase of water to its concentration relative to water in the gas phase. Its value is somewhat above 1.00, which it approaches with increasing temperature; Baumgartner and Kim (1990) measured values, compared them with compiled data and prepared a curve of a vs. temperature.
The conventional procedure in monitoring tritium oxide is to use dried silica gel from containers sealed by the manufacturer and dry the material at 100o C before use to remove any moisture that may have been adsorbed in the interim. The manufacturer (W. R. Grace & Co., Davision Chemical Div., Baltimore, MD 21203-2117) of the silica gel used in this study reported that the remaining water is at most 5.6% by weight, based on weight loss when heated to 950oC.
The tests in this study were performed with columns of silica gel weighing between 410 and 450 g. The silica gel and column are used routinely in environmental surveillance at the Savannah River Site (SRS). The amount of silica gel is selected to assure complete retention of water vapor during the 2-week collection periods from 3 m3 air. The amount of adsorbed water was estimated to range from about 14 g in winter to 64 g in summer.
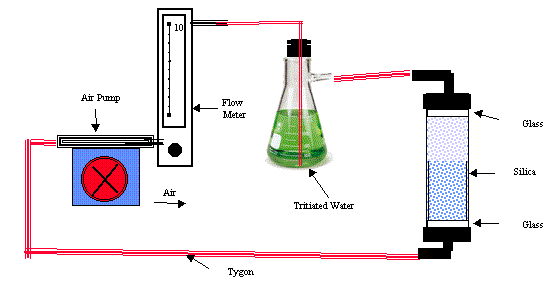
The silica gel was dried overnight at 100o C in open pans and placed in a column. Air was pumped through a tritiated water solution and the column in the closed circulating system shown in Fig. 1 until amounts of water ranging from 4% to 14% of the weight of silica gel were adsorbed. The silica gel was then transferred to a flask to recover water by distillation for tritium measurement. The tritium concentrations in the recovered water were compared with the initial concentration in the tritiated water solution. The ratio was corrected for the water vapor isotope effect both for the vapor carried in the air relative to the tritiated water and for the vapor distilled from the silica gel. The fraction of exchangeable water on silica gel inferred from these measurements was compared with the fractional water reported by the manufacturer and the weight loss fraction measured by thermogravimetry. The collection system then was operated in the environment of the Savannah River Site (SRS) to compare measured and corrected values under ambient conditions.
The silica gel used was Tel-tale, grade 42, which contains a colored moisture indicator. The plastic collection column is 5 cm i.d. and 30 cm long. Screw caps with 0.5 cm i.d. inlets, covered on the inside with glass wool, are at both ends. The column was weighed, filled with silica gel, and reweighed to determine the amount of silica gel.
The collector was inserted in the closed circulating system shown in Figure 1. A 140-ml solution of tritiated water at a concentration of 2 to 5 Bq/ml was placed in the bubbler flask. Air at 20 to 22oC was pumped through the tritiated water and the collector at a rate of 2 to 3 L/min. The system was operated for periods between 8 and 32 h so that the silica gel would adsorb amounts of water within the range of humidity expected in the environment throughout the year. The humidity in the inflow air was approximately 70%. The system was checked for leakage and modified until no further leakage was observed.
The tritium concentration in the flask was sampled in duplicate before starting the pump. Ten-ml aliquots were mixed with 10 ml scintillation cocktail and counted with an LS counter. The tritium counting efficiency (25%) and background (5-6 count/min) in the energy region used for tritium were measured periodically. Two 50-min counts were obtained per sample and averaged. The tritium net count rate per sample mass was calculated for comparative measurements, and the activity, for field measurements.
At the end of each run, the silica gel column was weighed to determine the amount of adsorbed water. The silica gel then was transferred from the column to a distillation flask or a cyclohexane azeotrope reflux flask and mixed thoroughly. The silica gel in the heating flask was heated to maintain it at about 160o C for 0.8 to 3 hours, depending on the amount of adsorbed water. For azeotropic distillation, 320 ml cyclohexane were added and the mixture was boiled at 80o C to collect water for 1 - 3 h, as indicated by the change in color from pink to blue of the silica gel, or until sufficient water had been collected.
The first 12 ml and the second 12 ml (or less, for low amounts of adsorbed water) of the distillate were collected. Aliquots of 10-ml (or if less, smaller aliquots diluted to 10 ml with non-tritiated water) were mixed with scintillation cocktail and counted with the LS counter. The two results were converted to tritium concentration and averaged.
The tritium fraction -- the ratio of tritium concentrations after desorption to before adsorption on the silica gel -- was calculated. The measured tritium fraction was corrected for the isotope effect on the basis of constructed curves of tritium concentrations in vapor relative to the initial tritium concentration in water. The percent exchangeable water was calculated from the corrected tritium fraction and the percent adsorbed water.
The standard deviation of the measurement was estimated from the total count of the samples, the background count, and the counting period. Also considered for contribution to uncertainty were the uncertainty of pipetting (0.5% estimate) and of weighing the tared column and the column with silica gel before and after water-vapor sorption (0.5 g estimate). On this basis, the coefficient of variation for the percent exchangeable water was estimated to be 0.08.
Sets of six 5-g samples of the silica gel were heated sequentially to various temperatures between 100o and 1050o C. After heating for 16 h at one temperature, the samples were cooled in a desiccator, weighed to observe the water loss as a function of temperature, and returned to the furnace.
A field collection system, consisting of a column filled with 415 to 420 g silica gel and an air pump, was placed in a conventional SRS air monitor housing located northwest of SRS just outside the boundary. It was operated for six consecutive 2-week periods beginning October 8, 1997. At the end of every period, the used silica gel column was replaced with a fresh column, and was taken to the laboratory where the silica gel was weighed, transferred to a distillation flask and mixed. A fraction of the adsorbed water was collected by azeotropic distillation and two aliquots were measured for tritium in an LS counter. The results were corrected for isotopic exchange and the isotope effect.
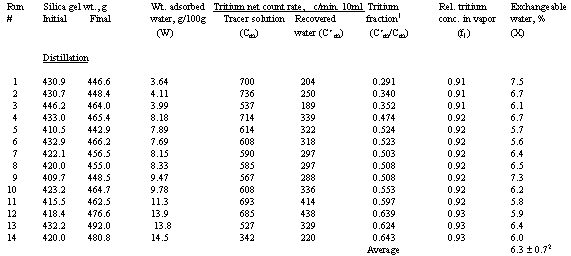
The tritium concentration data in Table 1 show that the values in the
desorbed water were well below those in the initial tracer solution, with a
trend of lower tritium fraction for less adsorbed water. The results suggest
dilution by exchangeable water, and the trend suggests that the percent
exchangeable water in the silica gel may be constant.
If exchange of tritiated water with non-tritiated water and hydroxyl groups is
complete, then the following tritium balance can be written:
C'(W + X) = C W (1)
where C is the initial concentration of tritium in adsorbed water and C' is the concentration of the desorbed water, both in count/min.ml; W is the amount of water adsorbed on the silica gel and X is the amount of exchangeable water in the silica gel before adsorption, both in g per 100 g dried silica gel or percent. The value of X is calculated with the equation by:
X = W [(C/C') - 1] (2)
Listed in Table 1 are the measured values Cm initially in the tracer solution and C'm in the distillate samples, which must be corrected for the isotope effect before equation (2) can be applied. The correction factor for the isotope effect for water vapor swept from the tracer solution at 20 to 22oC and adsorbed on the silica gel is defined as f1 = C/Cm. The correction factor for the isotope effect for adsorbed water distilled from the silica gel to measure tritium is defined as f2 = C'm/C'. Hence, the measured ratio Cm/C'm -- the inverse of the tritium fraction -- is converted to the ratio in equation (2) by
C/C' = (f1f2) Cm/C'm (3)
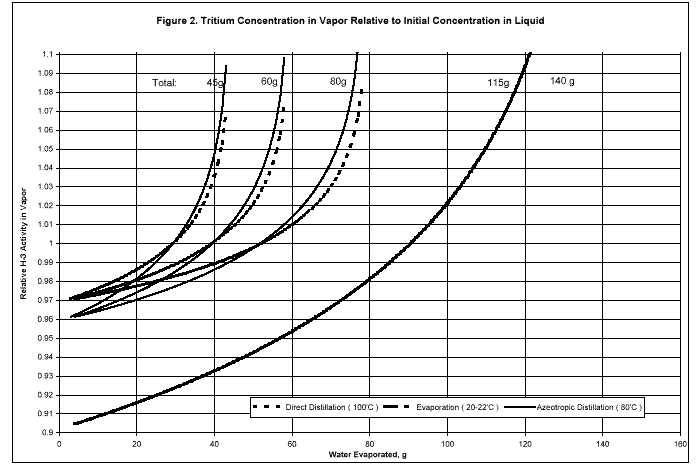
Point values for the isotope effect correction factor -- the tritium concentration in vapor relative to an initial concentration of 1.00 in water -- as a function of the amount of vaporized water are shown in Fig. 2. The curves are based on values for a of 1.11 at 20 to 22oC in sweeping water vapor from the tracer solution, 1.04 at 80oC in azeotropic distillation, and 1.03 at 100oC in direct distillation. The values were measured with tritium tracer at these temperatures (Rosson et al. 1998) and are consistent with published values (Baumgartner and Kim 1990).
Values of f1 averaged for the point values in Fig. 2 for each run between 0 g to the amount vaporized are listed in Table 1. Values of f2 averaged in the same way for distillate samples were found to be within 0.01 of 0.97 for azeotropic distillation and 0.98 for direct distillation in all runs.
The values of X calculated from the measured values in Table 1 according to equations (2) and (3) agree for both types of distillation (see Table 1). They yield an overall average Xa of 6.3%. The standard deviation for all 22 values is +/- 0.5%, and the standard error is +/- 0.1%.
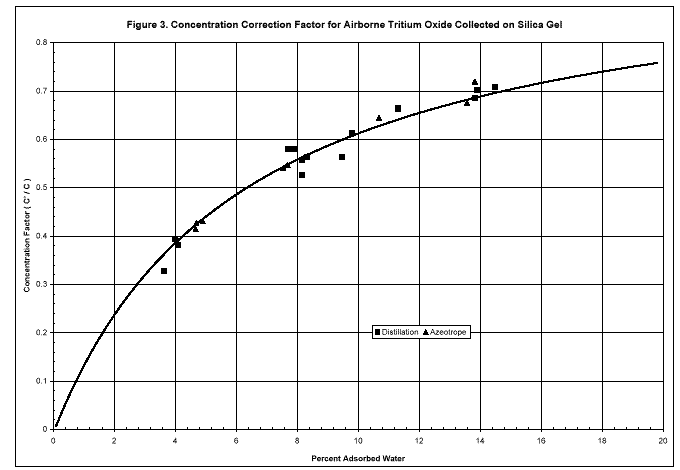
The curve of the isotopic exchange correction factor W/(W + Xa) plotted in Fig. 3 is consistent with the individual tritium fractions C'/C. All points lie within 8% of the curve. To correct a value C', it is divided by the point on the curve appropriate to the percent water adsorbed on the silica gel. The measured value C'm first is corrected for the isotope effect by dividing by 0.98 or 0.97, depending on whether direct or azeotropic distillation was used for recovering the adsorbed water for tritium counting. No correction is needed for the vapor pressure isotope effect when water vapor is collected on a monitoring column as long as all of the vapor is adsorbed.
Several tests that compared results for tritiated water recovered from silica gel that had either been mixed or not mixed in the distillation flask showed that the tritium concentration in desorbed water from the mixed samples was lower by 14% to 27%. Mixing apparently is needed to assure that the entire silica gel participates in isotopic exchange. Confirmatory tests with segmented columns indicate that isotopic exchange approaches the maximum value when the segment approaches saturation. It is incomplete in relatively dry segments until the dry silica gel is removed from the column and mixed with moist silica gel (Rosson et al. 1998).
The thermogravimetric results in Table 2 yield a silica gel weight loss between 100 and 1,050oC of 5.3 +/- 0.1%. This value is consistent with the manufacturer's report that the maximum water content observed when heating the silica gel from 100 to 950oC was 5.6%. Most weight was lost between 300 and 850oC.
The observed weight loss of silica gel generally is taken to be the amount of water lost, but is not necessarily the amount of exchangeable water. In the present case, some exchangeable water or hydroxyl groups could be retained by silica gel above 1,050oC; moreover, water loss can occur by
2 =SiOH = (=Si)2O + H2O (4)
where two hydroxyl groups available for tritium exchange in the silica gel yield only one water molecule distilled from it. Hence, the 6.3% exchangeable water and 5.3% weight loss are consistent with 4.3% water and 2.0% hydroxyl groups in silica gel. For other types of silica gel, the percent exchangeable water and weight loss may be different; and the percent exchangeable water could be less than the percent water loss because some forms of water or hydroxyl groups are unavailable for prompt tritium exchange.
The magnitude of the correction is indicated by a set of tests performed at a sampling location near SRS during six 2-week periods in October - December, 1997 (see Table 3). Collectors sampled about 3 m3 air at an average humidity of 6 to 17 g/m3. The corrected tritiated water concentration in air was 1.4-2.6 times as high as the measured value during this period, but the ratio would be expected to be less at an higher average humidity for the entire year. Non-use of the correction would explain why the annual averages of measured tritium concentrations in the environment of the Savannah Rive Site were reported to be lower by factors of 1.0-2.5 than concentrations calculated with a dispersion model from annual release data in 1985 to 1994 (Simpkins and Hamby 1997).
Tests with silica gel exposed to tritiated water vapor in a closed system show that the concentration of tritium in water desorbed by distillation is significantly less than it was before adsorption. The extent to which the tritium concentration is reduced decreases as more water is collected. These observations can be explained in terms of isotopic exchange of adsorbed tritiated water with non-tritiated water and OH groups associated with silica gel dried at 100o C.
For the silica gel under study, the exchangeable water content was found to be 6.3%. This result was obtained with tritium tracer solution for which the measured results had been corrected for the HTO/H2O vapor pressure isotope effect.
The amount of exchangeable water exceeds the weight loss to 1,050oC of 5.3% and the manufacturer's report of 5.6% maximum water content. The amount of exchangeable water need not match the weight loss because the exchange may occur in part with two hydroxyl groups that form one water molecule upon heating, or because additional water and hydroxyl groups may remain with the silica gel until it is destroyed at a higher temperature.
In view of these findings, it is recommended that the presented equation or graph be applied to measured tritium concentration for the studied silica gel to correct for dilution by isotopic exchange. Isotopic exchange correction factors for other types of silica gel can be obtained with similar tritium tracer measurements. The silica gel must be mixed before the adsorbed water is recovered, and a vapor pressure isotope effect correction must be applied for adsorption and desorption in the tracer test, and for desorption of monitoring samples.
This work was supported in part by the Westinghouse Savannah River Co. under ERDA Task Order 96-078. We thank Mr. Jeffrey Lahr for the thermogravimetric measurements.

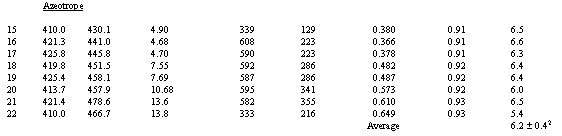
1. count rate of recovered water/count rate of tracer solution.
2. ± value is standard deviation for set of results.
Table 2. Silica Gel Weight Loss
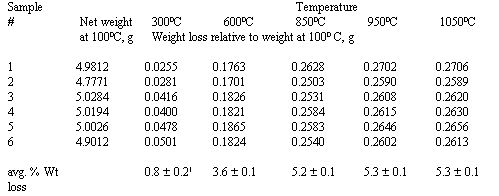
1. ± value is standard deviation
Table 3. Measurements of Airborne Tritium Oxide near the Savannah River Site
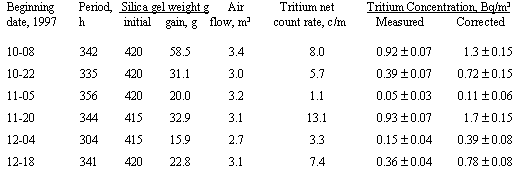
Note: The ± values are 1 standard deviations of counting.