
WSRC-TR-2001-00162
Evaluation of Cargo Leak Seal Product for Use in Tank 30 Cooling Coils
G. T. Chandler, C. F. Jenkins, J. I. Mickalonis, and D. A. Wilson
Westinghouse Savannah River Company
Aiken, SC 29808
This report was prepared as an account of work sponsored by an agency of the United States Government. Neither the United States Government nor any agency thereof, nor any of their employees, makes any warranty, express or implied, or assumes any legal liability or responsibility for the accuracy, completeness, or usefulness of any information, apparatus, product or process disclosed, or represents that its use would not infringe privately owned rights. Reference herein to any specific commercial product, process or service by trade name, trademark, manufacturer, or otherwise does not necessarily constitute or imply its endorsement, recommendation, or favoring by the United States Government or any agency thereof. The views and opinions of authors expressed herein do not necessarily state or reflect those of the United States Government or any agency thereof.
This report has been reproduced directly from the best available copy.
Available for sale to the public, in paper, from: U.S. Department of Commerce, National Technical Information Service, 5285 Port Royal Road, Springfield, VA 22161, phone: (800) 553-6847, fax: (703) 605-6900, email: orders@ntis.fedworld.gov online ordering: http://www.ntis.gov/support/ordering.htm
Available electronically at http://www.osti.gov/bridge/
Available for a processing fee to U.S. Department of Energy and its contractors, in paper, from: U.S. Department of Energy, Office of Scientific and Technical Information, P.O. Box 62, Oak Ridge, TN 37831-0062, phone: (865 ) 576-8401, fax: (865) 576-5728, email: reports@adonis.osti.gov
1.0 Summary
Pre-deployment and degradation testing of commercial leak seal products was performed to evaluate the potential for short-term repair of Tank 30 cooling coils. Four leak seal candidates were identified from a review of leak seal products and from previous testing for the Defense Waste Processing Facility. Seal-Upâ, a liquid glass metallic product manufactured by Cargo Chemical Corporation, was identified from the test results as the primary candidate to seal small leak sites. Pre-deployment testing was performed on test pipes with manufactured leak sites using a water flow loop system with nominal Tank 30 pressure (~50 psig). The maximum leak dimensions that the Cargo product was successful in sealing while the test piece was immersed in water was a slit, 0.016 inches wide ´ 0.291 inches long. This sized slit corresponded to a 1.34 gallon per minute leak rate at nominal Tank 30 cooling coil pressure. The maximum diameter leak hole that was successfully sealed with Seal-Upâ was 0.046 inches, which corresponded to a leak rate of 0.63 gallons per minute. The two lowest leak rates of the Tank 30 cooling coil assemblies are 0.07 and 0.9 gallons per minute.
Degradation testing included immersion and radiation testing of sealed pipe sections and fiber samples in simulated Tank 30 supernate. The Cargo product experienced degradation when exposed to these conditions. A Cargo seal in a test pipe remained viable for 50 days when exposed to Tank 30 supernate at 75-80°C with low-pressure cooling water at 27-35°C and included several salt/desalt cycles. A small leak (~0.03 gallons per minute) was observed after 23 days of testing. However, the leak self-healed when placed in a water flow loop with nominal Tank 30 pressure, which suggested that a Cargo seal may remain viable for >50 days under nominal pressure and Tank 30 conditions. A Cargo seal also remained viable after irradiation to 1.66E7 Rads, which is equivalent to approximately 2 years of service under expected Tank 30 dose rates.
The limited testing that has been performed does provide confidence that Cargo Seal-Upâ can provide a temporary seal for the Tank 30 cooling coils for small leaks. Attempts to inspect the leak sites of the Tank 30 coils were unsuccessful, so the location, geometry and size of the leak sites are unknown. A number of other factors can affect the success of the sealant. These factors include the turbidity of the chromate water; the degree of rusting, the curvature of the interior surface, fatigue loading of the coils; and the location, geometry, and size of the leak site(s).
Two additional tasks are being conducted to support this repair approach, although they are not discussed in this report. A compatibility analysis, which is being performed by Consolidated Storage and Transfer, includes an evaluation of the impact of the Seal-Upâ chemistry on the waste and waste processing. Also, a deployment system has been designed, fabricated and delivered by SRTC/EES. This system is capable of injecting a Cargo sealant mixture to individual Tank 30 coil assemblies.
2.0 Introduction
High Level Waste Tank 30 (TK30) is the concentrate receipt tank for the 3H Evaporator. TK30 contains five cylindrical deployable type cooling coil assemblies. A chromate cooling water is circulated through the coils to cool the contents of TK30. The coils are 2-inch diameter Schedule 40 carbon steel pipe. In November 2000, all five cooling coil assemblies leaked within one week of each other after the waste level was reduced. Two of the TK30 assemblies, B9 and B6, have relatively high leak rates of 66 and 23 gallons per minute (gpm), respectively. Assembly B4 has a leak rate of 6 gpm, and assemblies B10 and B2 have relatively small leak rates of 0.9 gpm and 0.07 gpm. Inspection efforts were unsuccessful in locating the leak sites.
The Savannah River Technology Center (SRTC) was requested to evaluate commercial leak seal products as a potential short-term repair approach for at least some of the TK30 cooling coils. SRTC quickly mobilized a program to assess the feasibility of this approach and to determine application requirements including the design and assembly of a deployment skid in response to a HLWM Technical Task Request [1]. Seal-Upâ, a liquid glass metallic product manufactured by Cargo Chemical Corporation, was chosen as the primary candidate from a field of four products. The other candidates did not work as effectively as Cargo in the screening tests performed in this task. The test program included chemical and morphological characterization of the Cargo sealant, simulated cooling coil testing with manufactured leak sites, and assessment of chemical and radiation resistance. This topical report discusses results for the Cargo product only. A comprehensive report detailing the complete program will be issued at a future date.
The planned testing was reviewed per the "Savannah River Technology Center Conduct of Research & Development (U)" for proper research [2]. Data for this project was logged in either notebook WSRC-NB-97-0036 [3] or EES Job Number 22810 [4].
3.0 Characterization
The Cargo leak seal product was characterized in the initial fluid form and as the as-formed seal. The chemical and morphological analysis techniques included Ion-Coupled Plasma Emission Spectroscopy (ICPES), Ion Chromatography (IC), X-ray Diffraction (XRD), Fourier Transform Infrared Spectroscopy (FT-IR), and Scanning Electron Microscopy (SEM) with Electron Dispersive X-ray Spectroscopy (EDS).
3.1 Chemical Analysis
The compositional information released by the manufacturer is given in the Material Safety Data Sheet (MSDS). The Cargo Seal-Upâ MSDS (# 3076-1) shows that sodium silicate, which is referred to as "liquid glass", and copper fines (10 wt % max) are the principal components. From a discussion with a chemist from the manufacturer, other constituents in the product include oxidizers and corrosion inhibitors [5].
The SRTC analytical results of the as-received product were in basic agreement with the composition indicated in the MSDS. Seal-Upâ was alkaline with a measured pH of 11.7. The ICPES analysis was performed on both the as-received product and the filtered liquid. Filtering was performed to remove most of the observed particulate. The as-received product consisted primarily of sodium, silicon, copper, aluminum, and iron, which analyses are given in Table 1. The aluminum may be acting as the oxidizer to which the Cargo chemist referred. Much smaller quantities of the metallic elements remained after filtering which indicated their presence as solids components. From IC, the principal anion found was nitrite (1900 ppm), which may be associated with the corrosion inhibitor.
Table 1. ICPES Analysis For Cargo Seal-Upâ Metallic Products
|
Element |
Concentration (ppm) |
|
|
As-Received |
Filtered |
|
|
Na |
57,000 |
71,000 |
|
Si |
103,700 |
131,000 |
|
Al |
800 |
500 |
|
Cu |
1,500 |
500 |
|
Fe |
300 |
100 |
The compositional analysis by FT-IR showed that the filtered product was made from a mixture of liquid polymers. The FT-IR spectrum for Seal-Upâ was diffuse without well defined peaks that would correspond to specific components. There were, however, wide peaks that corresponded to hydroxyl and carboxyl absorbances. These liquid components are believed to be the precursors for the fibers that are formed during the sealing process. These fibers become incorporated at the leak site as part of the seal. These fibers were analyzed by XRD and were shown to be cellulose. Aluminum and copper particulates were trapped between these fibers as verified with SEM/EDS analysis.
3.2 Morphology Of The Cargo Sealant
The morphology of the Seal-Upâ fibers was analyzed in several ways. The samples included as-received solids, which were collected on filter paper; filtered solids from the pressurized flow loop, a seal made in the pressurized flow loop, and seals and fibers that were exposed during the degradation testing. Test seals were made in steel pipe sections having predrilled holes and slits as described in Section 4, "Simulated Cooling Coil Testing". These samples were examined using the SEM/EDS.
The Seal-Upâ fibers were thin wall tubes, transparent or translucent, and predominantly beige or light tan in color. Figure 1 is a SEM photomicrograph of the Seal-Upâ fibers. The diameters ranged from about 6-20 microns (approximately 0.00024-0.00080 inches). Most fibers averaged 11-17 microns in diameter and had lengths less than one inch. The solid polymer fibers were not present in the starting material, but developed as the compound is applied. It appeared that the fibers would grow larger with time.
The sodium and copper contents of the seals were found to vary at the leak sites. These elements are part of the sodium silicate water glass and the copper fines described in the MSDS for the sealant. Both were found in the seal. The Cargo chemist stated that the copper particles function as a heat sink [5]. Additional work would be needed to determine more precisely the mechanism of seal formation.
The Cargo sealant is designed for application in internal combustion engine cooling systems. Under most circumstances, such seals are not exposed to an aggressive liquid, but are exposed to a pressurized gaseous environment. However, the waste tank cooling system leaks are in piping that is immersed in the liquid waste. Exposure tests to water and a simulated waste were required. The waste exposure was not expected to impact the formation of fibers and the seal since the Cargo product is alkaline.
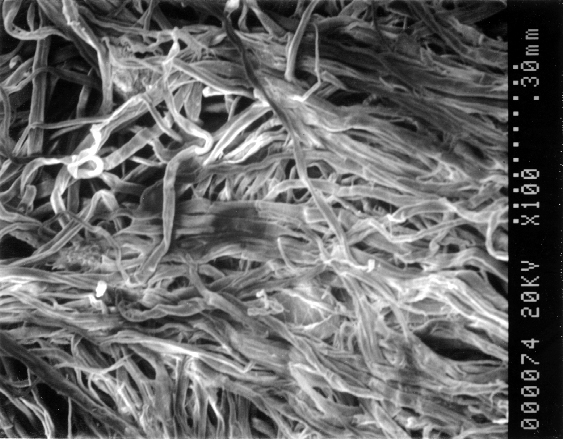
The water and simulated waste exposure resulted in dissolution and reduction of the water glass component of the sealant material (sodium silicate) as shown in Figure 2. The degradation in the waste appeared more rapid than in the water. In both cases, the fibers remained after the exposure, though they became matted or pressed closer together as the fibers collapsed and the loose stacks were compressed.
3.3 Proposed Sealing Mechanism
The following mechanism for sealing leaks with Seal-Upâ is based on the chemical and morphological characterization and the degradation testing. As a result of thermally assisted formation, polymeric fibers, which consist of cellulose, trace the flow pattern of the water. The polymer is initially in solution as part of the liquid component of the sealant. At a hole where the solution changes direction due to the leak, sealant constituents follow this new direction. The fibers tend to align with the flow, but as the flow pattern compresses and then expands at the opening, the tails become entangled on the inside to develop a mass of fibers. The copper, aluminum and sodium silicate, constituents of the sealant, fill into the interstices of this mass to complete the seal. The mechanism is similar to that previously described for liquid seal product that was used in the repair of a water leak in the DWPF melter system [6].
4.0 Simulated Cooling Coil Testing
The simulated cooling coil testing consisted of two types of tests. One type evaluated the sealant capabilities at the nominal pressure and flow conditions of the TK 30 cooling coils. This test was conducted in a pressurized flow loop and was used to evaluate seal formation and integrity with the outer portion of the leak site exposed to water under nominal flow conditions. The other test was used to evaluate the integrity of a sealed pipe under chemical and thermal conditions, which simulated the environmental conditions of the TK 30 cooling coils.
4.1 Pressurized Flow Loop Results
Tests were performed to evaluate the sealing capability of Seal-Upâ for carbon steel pipe sections with machined leak sites that were varied by geometry and size. The pipe sections were 0.5-in Schedule 40 pipe and approximately six inches long. Since the geometry and size of the TK 30 leak sites are unknown, two geometries and sizes were evaluated. The leak sites, which were holes and slits, had leak rates ranging from 0.63 to 1.44 gpm with TK 30 nominal pressure. Holes were made by drilling and slits were machined by electrical discharge machining (EDM). Each pipe section had both a slit and hole. The standard-sized leak sites were a 0.046-in diameter hole (0.63 gpm) and a slit with dimensions of 0.016 in wide by 0.291 in long (1.34 gpm). The pipe sections had either air or water exposed on the exterior, while a solution of Seal-Upâ and water flowed through the interior. These pipe sections with sealed leak sites were also used in the environmental flow loop.
The test loop, which is shown in Figure 3, was a simple system consisting of a pump that provided a leak rate through a pipe section containing the machined through-wall leak sites. The pipe section was connected on the discharge side of the pump after a flow meter. The flow was maintained at approximately 6 gpm. After exiting the pipe section, the water flowed to a 2.5-gallon water reservoir, which supplied heated water to the pump. A polyvinyl chloride trough below the test pipe section recycled any leaking water back to the reservoir. Pressure and temperature were monitored using analog gauges. Pressure was maintained at 50 psig (TK 30 nominal pressure). The temperature was ramped from 115 to 160° F (46-71°C) during testing. The total liquid volume for the loop was 4 gallons, which included 16 ounces of Seal-Upâ.
Seal-Upâ formed a seal in the standard slit in 4 minutes and in the standard hole in 20 minutes when exposed to air. The time for seal formation was longer for standard-sized leak sites when the pipe was exposed to water. The slit and hole sealed in 20 and 40 minutes, respectively. The sites remained sealed for the length of the test; the longest test time was 120 minutes. Seal-Upâ, however, did not seal larger leak sites with water exposure. The leak rate showed no apparent decrease for a hole with a 0.062-in diameter (0.72 gpm) or a slit with dimensions of 0.046-in width by 0.101-in length (1.44 gpm).
From visual observations during testing, the seal was found to form by filling and protruding through the leak site and then coalescing outside the leak site. The other candidate sealants that incorporate fibers did not appear to form as large a mass on the outside of the pipe. The Seal-Upâ fibers, however, slowly filled the leak site and occluded the flow as opposed to piling up quickly at the site. This piling might prevent fiber coalescence outside the leak site.
Several tests were performed to evaluate the impact of fibers that might remain in the cooling coils after sealing was complete. As discussed above, the leak sites remained sealed when not exposed to an aggressive environment (i.e., water versus a waste solution). The free flowing fibers, in this case, either maintained the seal or may have had no impact. In an aggressive waste solution, the seal in a cooling coil was hypothesized to deform and leak or possibly additional leak sites would form. For testing this scenario, preformed fibers were injected into the loop with a pipe containing both the standard-sized leak sites. The slit sealed in 40 minutes at 70°C. The hole, however, did not seal. When the flow direction was reversed, the hole was sealed in 90 minutes. A large build up of fibers at the slit had prevented the free flowing fibers from sealing the hole. The hole and slit were 0.75 inches apart.
Several different approaches were attempted to assess the requirements for seal formation using standard-sized leak sites. Flow and heat were found to be necessary for the fibers to form in the pressurized flow loop. Fibers did not form in a laboratory beaker, which contained a stirred solution of Seal-Upâ. At low temperatures (30-40°C), the solution clouded; at higher temperatures (60-70°C) cotton-like clumps formed.
Operational parameters were to be investigated as time permitted in order to optimize the Cargo seal formation in the cooling coil. These parameters included the need for preheating the water, ultimate water temperature, degree of agitation, and addition rate of the sealant. Since time was not available, some of these parameters were discussed with the manufacturer. Their recommendation was to follow the directions given on the bottle as closely as reasonable [5].
4.2 Environmental Flow Loop Results
The environmental flow loop test was conducted to perform longer term testing with coolant flow and simulated TK 30 conditions to evaluate the impact of salt and supernate exposure on seal integrity. The setup consisted of a test pipe section connected to a low-pressure water coolant flow loop with the outside of the pipe section exposed to TK 30 simulated waste. The test section contained standard-sized leak sites, which had been previously sealed with Seal-Upâ in the pressurized flow loop. The coolant water was controlled between 27 and 35°C using a recirculating water bath to simulate cooling coil conditions. A glass vessel with a reflux condenser was used to reduce evaporation of the TK 30 simulated waste. The initial composition of the TK 30 simulated supernate is shown in Table 2. The vessel was heated using a hot plate and heat tape (when needed) to maintain the simulated supernate between 68 and 75°C. The test setup is shown in Figure 4.
Table 2. Supernate Chemistries For Seal-Upâ Degradation Testing
|
Component |
Concentrations (M) |
|
|
Tank 30 |
Chromate Water |
|
|
NaOH |
5.0 |
0.0001 |
|
NaNO3 |
3.0 |
-- |
|
NaNO2 |
1.7 |
-- |
|
Na2CrO4 |
-- |
0.0043 |
The first test was conducted in several phases. During the first eight-day phase, sodium nitrate was added periodically to obtain a salt concentration at which a salt layer formed on the pipe section. The second phase was conducted for four days to allow a complete salt layer to form. Figure 5 shows several sequential photographs taken during salt layer formation. During the third phase, desalting of the pipe was performed by shutting off coolant flow to the pipe. The supernate temperature, however, spiked to 100°C. The sample pipe leaked after the circulating bath was turned on and the leak sites did not reseal. When the pipe was placed back in the pressurized flow loop, the leak sites also did not reseal.
For the second test, a new sealed pipe section was placed into the loop with a fresh batch of supersaturated TK 30 simulated supernate. The exposure sequence and test results are summarized in Table 3. The test was operated for 23 days until a small leak was detected. Three salt/desalt cycles were performed over the first ten days without any leaking or temperature spikes. During the subsequent thirteen days, a salt layer was maintained continuously on the pipe section until a small leak was detected (~<0.01 gpm). Some salt dissolution occurred during the last few days probably due to dilution of the supernate.
The pipe section was placed in the pressurized flow loop with fresh water to evaluate the degree of leaking and the self-repairing capability of the seal. Only slight weeping was observed at the hole, while leakage was observed at both ends of the slit. The leak rate from the failed seals at nominal pressure was measured to be approximately 0.03 gpm. Leaking from a failed sealed at a slit typically occurred at the short ends of the slit. These ends had sharp edges on the interior, which resulted from the machining.
The seals were observed to heal within approximately one minute to one drop every 20 seconds. Recurrence of the leak and then rehealing over short durations were observed at the slit several times during approximately two hours of testing. The leak rate was approximately 1 drop/min at the end of testing. After removal from the flow loop, the interior of the pipe was inspected and found to have approximately 50% less product below the slit. The hole had little apparent loss of product.
The test piece was placed back into environmental flow loop with the TK 30 simulated waste. The loop conditions were the same as used earlier. A salt layer formed immediately and was maintained for an additional 23 days. This time length of continuos operation for the cooling coil was taken as representative of the tank farm.
Table 3. Long-Term Environmental Flow Loop Results
|
Test |
Salt Layer |
Solution |
Comments |
|
10 |
3 salt/desalt cycles |
<80 |
No leaking |
|
13 |
Continuously maintained |
75 |
No leaking |
|
1.5 |
Continuously maintained |
75 |
<0.01 gpm leak |
|
2.5 |
Slowly dissolved |
75 |
No coolant flow |
|
8* |
No salt exposure |
No supernate exposure |
Pipe tested in nominal pressure flow loop with water; 0.03 gpm leak at slit, leak healed; pipe maintained wet in Water/Seal-Upâ solution before and after nominal pressure testing |
|
23 |
Continuously maintained |
70-75 |
No leaking |
* Pipe section was transported between loops in water containing some Seal-Upâ product
5.0 Degradation Testing
Testing was performed to evaluate susceptibility of the candidate sealants to degradation under TK 30 conditions. The testing has shown that the sealants are affected by both chemical and radiation exposure. The Cargo sealant showed the best resistance under the test conditions compared to the other candidates. Chemical and radiation exposure testing was performed on sealed pipe sections and on isolated sealant fibers obtained from the pressurized flow loop.
5.1 Stagnant Chemical Testing – Sealed Pipes
Scoping tests were initially performed on sealed pipes exposed to TK 30 simulated supernate under non-flowing conditions. Pipe sections containing standard-sized leak sites were sealed under air or water exposure (on exterior of pipe). The pipe sections were then immersed in TK 30 simulated supernate at 80°C for 7-10 days. The pipe sections were then removed and checked for leakage under a static head pressure. The Cargo test piece showed no leakage from the hole or slit. The Cargo test pieces were then tested in the pressurized flow loop with external fresh water. Only minor weeping was observed at the hole; however, the slit was found to be leaking at one end. The leak stopped after approximately one minute with only minimal subsequent weeping.
5.2 Chemical Testing - Fibers
Chemical exposure tests were performed on fibers that were filtered from the water and Seal-Upâ mixture used during the pressurized flow loop testing. The filtered fibers were rinsed, dried and weighed prior to testing. Samples of the Seal-Upâ fibers were exposed to the TK 30 simulated supernate and chromate water at both 80 and 90°C. The chemical compositions of the solutions are given in Table 2. For this test, a fiber sample was placed in a centrifuge tube (glass or Teflon) and weighed. An initial weight of the centrifuge tube was also taken. Each week the samples were weighed using the following procedural steps: A) Centrifuge the tubes to push fibers to bottom, B) Decant the solution, C) Rinse with distilled water, D) Centrifuge, E) Decant rinse water, F) Dry in oven at test temperature, and G) Weigh samples.
Weight change results from the fiber tests are shown in Figure 6. The Seal-Upâ fibers lost weight in all the test solutions. In the chromate water, a 23 wt% decrease was observed after the first week of exposure at 80 and 90°C. No appreciable weight loss was observed in subsequent exposures up to 3 weeks. A 60% weight loss was observed when the sample was exposed in the TK 30 simulated waste for 4 weeks at 80°C. A significantly higher weight loss of 90% was observed after 4 weeks of exposure at 90°C. A fairly constant weight loss rate was observed at both temperatures over the 4 weeks of exposure. These results indicate that a seal in the cooling coil would have a greater degradation when exposed to the waste (without an exterior salt layer formed) than from the chromate water.
5.3 Radiation Testing
Radiation testing was performed to evaluate the effect of radiation on degradation of the seal and to determine the capability of the product to self-repair after exposure. A sealed pipe section immersed in TK 30 simulated supernate was irradiated in a cobalt-60 gamma source at 1.04 E6 rad/hr and approximately 50°C. The inside of the pipe was filled with a mixture of Seal-Upâ and water. The pipe was subjected to four consecutive 4-hour exposures for a total dose of 1.66E7 Rads, which is equivalent to 2 years of irradiation in Tank 30 assuming a dose rate of 1000 rad/hr. After each 4-hour exposure period, the pipe section was inspected for leakage under static head pressure and then tested in the pressurized flow loop. Table 4 summarizes the results. As shown, the seal showed no leakage after a 6-month equivalent exposure. Only a small drip or minor leakage was observed in subsequent exposures up to 2 equivalent years.
Another pipe was subjected to a much higher total dose of 1.0 E8 Rad, which is equivalent to approximately 10 years of service. The seal showed no leakage under static head pressure after a total dose of 5.0E7 Rad. However, after a total dose of 1.0E8 Rad, leakage was observed under static head pressure. The pipe was then inserted in the pressurized flow loop with only water in the system. The seal did not self-repair even though fibers remained in both the slit and hole, which reduced the leak rate from unsealed sites.
Table 4. Leak Detection After Radiation Exposure
|
Order |
Dose |
Cumulative |
Static Leak |
Dynamic Leak |
|
1 |
4.16 E6 |
4.1 E6 |
No |
No |
|
2 |
4.16 E6 |
8.3 E6 |
No |
Slit – 1 |
|
3 |
4.12 E6 |
12.4 E6 |
No |
Slit – 0.67 |
|
4 |
4.12 E6 |
16.6 E6 |
No |
Slit – 0.25 |
6.0 Discussion
The SRTC testing of the Cargo leak seal product has shown that under simulated TK 30 conditions there is a reasonable chance of success. Upon addition to a circulating water system, Seal-Upâ starts to form cellulose fibers. These fibers are drawn to the leak site as they circulate in the system. The complete mechanism of sealing is unknown; however, the copper particles were found to be present with the fibers. The sodium silicate, which is the major component of Seal-Upâ is also incorporated in the fiber agglomeration. When the leak site is exposed to air, the silicate will typically form a crystallized shell on the exterior. This shell is one part of the overall seal. The silicate may also act as a corrosion inhibitor for the metallic components of the system.
Testing does show that the Cargo product is susceptible to degradation under TK 30 supernate conditions. However, the environmental flow loop testing which was an attempt to closely mimic coil conditions, indicated that the seal was maintained for 50 days. Fiber immersion testing indicated that a significant amount of dissolution occurred in a relatively short time period. However, salt formation and coolant flow, as tested in the environmental flow loop, may serve as a barrier for accelerated degradation of the seal. When some leaking does occur, testing in the pressurized flow loop showed that the seal heals by the shifting the buildup of product located outside the leak site.
These results collectively suggested that a Cargo seal would remain viable for longer periods when exposed in TK30. The uncertainty, however, is dependent on the response of several factors including the turbidity of the chromate water; the degree of rusting, curvature of the interior surface, fatigue loading coil supports, and the location, geometry, and size of the leak site(s).
7.0 Conclusion
The testing that was performed by SRTC provides confidence that Seal-Upâ can provide a temporary seal for the leaking TK 30 cooling coils. The maximum leak site area that the Cargo product was successful in sealing while the test piece was immersed in water was a slit, 0.016 inches wide by 0.291 inches long, which corresponded to a 1.34 gpm leak rate at nominal TK 30 cooling coil pressure. The maximum diameter hole that could be successfully sealed with Seal-Upâ was 0.046 inches, which corresponded to a leak rate of 0.63 gallons per minute. The two lowest leak rates of the TK 30 cooling coil assemblies are 0.9 and 0.07 gallons per minute.
The Cargo product did experience degradation when exposed to simulated TK 30 conditions. A Cargo seal in a test pipe remained viable for 50 days when exposed to TK 30 supernate at 75-80°C with low-pressure cooling water at 27-35°C with several salt/desalt cycles. A small leak (~0.03 gallons per minute) was observed after 23 days of testing. However, the leak self-healed when placed in a nominal Tank 30 pressure water flow loop, which suggested that a Cargo seal may remain viable for >23 days under nominal pressure and Tank 30 conditions. A Cargo seal also remained viable after irradiation to 1.66E7 Rads, which is equivalent to approximately 2 years of service under expected Tank 30 dose rates.
The chance of success in the TK30 cooling coil is dependent on the response of several factors. These factors include turbidity of the chromate water; the degree of rusting, curvature of the interior surface and fatigue cycling of the coils; location, geometry, and size of the leak site(s). At this time, the impact of these factors is unknown for the seal formation and integrity.
8.0 Acknowledgments
The authors wish to acknowledge the conscientious efforts of Chris Beam, Tony Curtis, Karen Hicks, Dan Krementz, and Elise LaBord toward the successful completion of this task.
9.0 References

Figure 1. SEM photomicrograph of filtered Seal-Upâ fibers
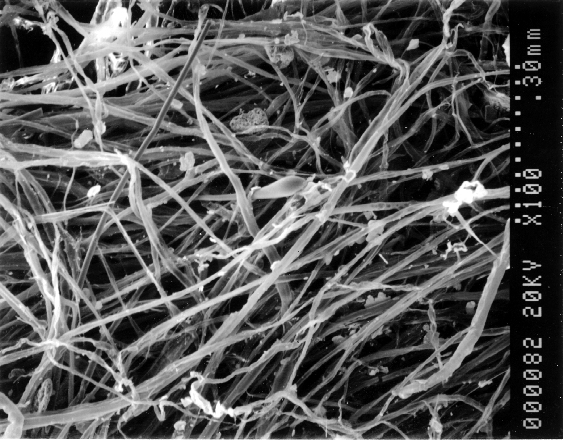
(A)
(B)
Figure 2. SEM photomicrographs of Seal-Upâ
fibers exposed to A) water
and B) sodium hydroxide solution

(A)

(B)
Figure 3. Pressurized Flow Loop: A) Complete system, B) Trough with pipe section

Figure 4. Environmental Flow Loop Test Set Up






Figure 5. Salt Layer Formation In Environmental Flow Loop
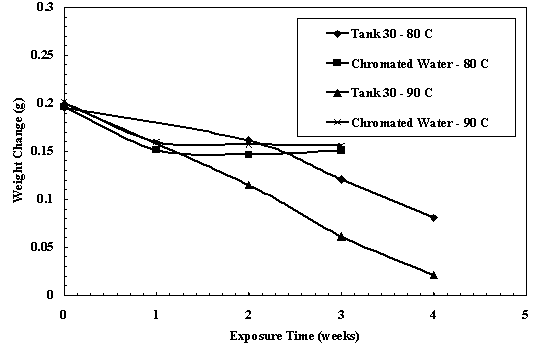
Figure 6. Weight Change Of Seal-Upâ Fiber In Chemical Degradation Tests