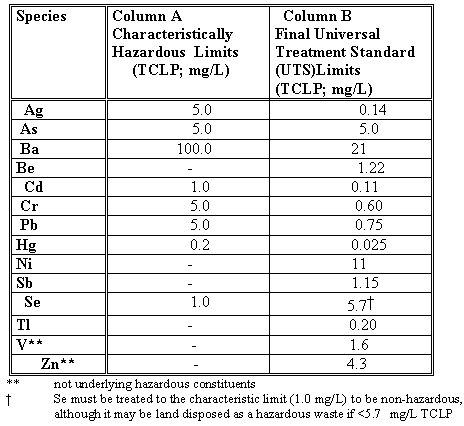
Table I. Environmental Protection Agency Concentration Limits for
Inorganic Constituents of Hazardous Wastes
WSRC-MS-2000-00195
Mining Industry Waste Remediated for Recycle by Vitrification
C.M. Jantzen, J.B. Pickett and R.F. Schumacher
Westinghouse Savannah River Company
Aiken, SC 29808
This report was prepared as an account of work sponsored by an agency of the United States Government. Neither the United States Government nor any agency thereof, nor any of their employees, makes any warranty, express or implied, or assumes any legal liability or responsibility for the accuracy, completeness, or usefulness of any information, apparatus, product or process disclosed, or represents that its use would not infringe privately owned rights. Reference herein to any specific commercial product, process or service by trade name, trademark, manufacturer, or otherwise does not necessarily constitute or imply its endorsement, recommendation, or favoring by the United States Government or any agency thereof. The views and opinions of authors expressed herein do not necessarily state or reflect those of the United States Government or any agency thereof.
This report has been reproduced directly from the best available copy.
Available for sale to the public, in paper, from: U.S. Department of Commerce, National Technical Information Service, 5285 Port Royal Road, Springfield, VA 22161, phone: (800) 553-6847, fax: (703) 605-6900, email: orders@ntis.fedworld.gov online ordering: http://www.ntis.gov/support/ordering.htm
Available electronically at http://www.osti.gov/bridge/
Available for a processing fee to U.S. Department of Energy and its contractors, in paper, from: U.S. Department of Energy, Office of Scientific and Technical Information, P.O. Box 62, Oak Ridge, TN 37831-0062, phone: (865 ) 576-8401, fax: (865) 576-5728, email: reports@adonis.osti.gov
Abstract
Characteristically hazardous waste water treatment sludges from a U.S. mining company were considered a long term liability since stabilization via a cement wasteform would not provide the stringent leachant concentrations for an EPA acceptable recycle product. Vitrification of the sludges into three different types of glass at elevated temperatures provided recyclable products. The use of treated materials containing hazardous metals has been previously considered by the EPA for residues remaining from High Temperature Metal Recovery (HTMR) operations. These treated materials could be used for recycling as (1) covered sub-base materials (e.g., in construction of paved roads, parking lots, and driveways), (2) additive ingredients in cement or concrete/asphalt mixtures, (3) top grade or surfacing materials, e.g., in construction of roads, parking lots, and driveways (glassphalt or glasscrete), and as anti-skid/de-icing materials. The glass waste forms provide a 87-93% volume reduction compared to alternative stabilization in cement and provide for recycle.
Introduction
Raw materials taken from the earth to produce a wide variety of products and processes must be disposed of safely back into the earth once declared a waste. The only other option is remediation for recycle into new products or new end uses. Technologies have been developed by the US Department of Energy’s (DOE) Westinghouse Savannah River Technology Center (SRTC) to convert a wide variety of hazardous wastes to a solid stabilized glass via the process of vitrification. This vitrification technology can render hazardous wastes to be non-hazardous and/or convert non-hazardous sludges, asbestos, etc. into recyclable products or reusable raw materials.
Vitrification
Vitrification atomistically bonds any hazardous species in a solid glassy matrix providing for recycle of the end product under certain conditions. The final waste form or product is, therefore, very durable and environmentally stable over long time durations. Vitrification processes are flexible enough to handle process chemistry variations and can accommodate dry or wet waste/recycle materials. The U.S. EPA has declared vitrification the Best Demonstrated Available Technology (BDAT) for high-level radioactive waste [1] and produced a Handbook of Vitrification Technologies for Treatment of Hazardous and Radioactive Waste [2].
Vitrification produces large volume reductions, e.g. up to 97%. Vitrification can use cheap sources of glass forming agents, e.g. sand, soil, mill tailings, crushed reagent bottles, beer bottles, broken window glass, etc. Compact and/or transportable melters (capable of vitrifying about 1.8 tons/day of wet feed or 3.5 tons/day of dry feed) technology minimizes costs [3-4]. Vitrification is cost effective on a life-cycle basis compared to other stabilization technologies.
Environmental Protection Agency (EPA) Regulations
Wastes that Fail the U.S. Environmental Protection Agency (EPA) Characteristically Hazardous Leaching Procedure (TCLP) for any of the inorganic species listed in Table I (Column A) are considered characteristically hazardous (Organic species are also listed by EPA in regulation 40CFR268.40) but organic species are volatilized at the high temperatures used in vitrification and are, therefore, not discussed further.). Prior to May 28, 1998 characteristically hazardous wastes could be treated so that they would meet the TCLP leachate levels given in Column A. The U.S. EPA promulgated a regulation on May 28, 1998 that characteristically hazardous wastes must be treated to the Universal Treatment Standards (UTS) (Federal Register, V.63, #100, p.28748-9) shown in Table I (Column B).
Case Study
A U.S. Mining company ran a waste water treatment facility that produced 3000 tons of wet sludge waste per year. The raw sludge failed TCLP for Cd and was, therefore, considered characteristically hazardous per column A in Table I. The remediation being used for this Cd containing sludge was to mix the sludge with cement. The cured sludge-cement mixture passed TCLP at the levels given in Table I Column A. However, the sludge-cement remediation was problematic for the following reasons:
A feasibility study was initiated to see if the waste water treatment sludge could be remediated by vitrification, rendered non-hazardous by EPA guidelines, and thus recycled instead of being declared a waste. The feasibility study included the following:
Table I. Environmental Protection Agency Concentration Limits for
Inorganic Constituents of Hazardous Wastes

Analysis of the Sludge and Mill Tailings
The raw waste was analyzed by x-ray diffraction after drying at a variety of temperatures. Wet chemical analysis was performed in duplicate after the sludge was dried at 105°C. It was determined that the waste contained 13.3 wt% solids and 86.7 wt% H2O after drying at 105°C (Table II). The dried sludge was comprised of CaCO3 (calcite), (Na,K)3(Mn,Mg,Fe,Al)5Si8O22(OH,F)2 (Kozulite), and ZnFeCrO4 (Spinel) by x-ray diffraction analysis. The remaining x-ray diffraction analyses demonstrated that the sludge lost an additional 2.8 wt% hydroxides and carbonates from the kozulite and calcite phases upon calcining at 1050°C, e.g. ~1.5 wt% OH- from the hydroxide minerals present, plus ~1.5 wt% from the carbonate species and nitrates present. Whole element chemistry (Table II) indicated that the waste contained only 0.12 wt% CdO but also contained 25.64 wt% ZnO, and 7.01 wt% CaO. The waste also contained 4.79 wt% SO4 from unknown origin. The speciation of the oxidized Fe2O3 and reduced FeO and the oxidized CuO and the reduced Cu2O was determined from the measured Fe+2/å Fe ratio of 0.054 and the known EMF series for iron and copper bearing waste glasses [5]. Likewise, the measured Fe+2/å Fe indicated that all of the Mn species in the waste would be present as MnO and not MnO2 or Mn2O3.
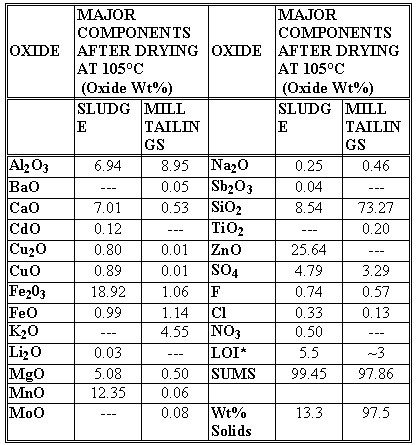
Mill tailings provided by the mining company were analyzed in the same fashion to examine their usage as glass forming additives. X-ray diffraction analysis indicated that the mill tailings were primarily SiO2 (quartz) with minor amounts of KAl2(Si3Al)O10(OH,F)2 (muscovite), KAlSi3O8 (microcline feldspar), and CaSO4•2H2O (gypsum). The duplicate wet chemical analyses indicated that the mill tailings contained ~73-75 wt% SiO2, ~9 wt% Al2O3, ~4-5 wt% K2O which are all excellent glass forming oxides. However, the mill tailings contained CaSO4•2H2O (gypsum) which comprised 3.29 wt% SO4. The speciation of the oxidized Fe2O3 and reduced FeO and the oxidized CuO and the reduced Cu2O was determined from the measured Fe+2/å Fe ratio of 0.54 and known EMF series for iron and copper bearing waste glasses [5]. Similar to the sludge analysis, the measured Fe+2/å Fe indicated that all of the Mn species in the waste would be present as MnO and not MnO2 or Mn2O3.
The waste sludge was successfully made into three different types of glass at a variety of melt temperatures (Table III):
Of the twelve glass melts studied, nine made excellent waste glass. Most of the glass formulations were in the soda-lime-silica system since borosilicate glasses are not tolerant of high concentrations of divalent cations such as Ca+2, Sr+2, Zn+2, Pb+2: the borosilicate glasses separate into two immiscible phases [6-7]. This type of phase separation can compromise the processing, durability, and overall stability of the vitrified product and should be avoided. In addition, soda-silica glass is known to be extremely tolerant of high zinc (Zn) [8] and other heavy metals. Indeed, SLS glasses were fabricated from high Zn containing incinerator ash and off-gas blowdown at SRTC [9].
Nine of the 12 glasses formulated were SLS glasses (Table III) due to the relatively
high concentration of CaO and ZnO in the waste (Table II). Two of the 35 wt%
loaded glasses (CC-1 and CC-4) were made with 50 wt% mill tailings and an additional
15 wt% fluxing agents, e.g. calcium carbonate (CaCO3), sodium
carbonate (Na2CO3), and lithium carbonate
(Li2CO3). The fluxing agents are added
to glass formulations in order to keep the melt temperatures low and retain
hazardous components that can sometimes be volatile at higher melt temperatures.
Glass CC-1 was melted at 1350°C and glass CC-4 was melted at 1250°C. The glass
melted at the lower temperature (CC-4) in the presence of additional fluxing
agent (Li2CO3), contained undissolved
waste solids, primarily ZnFeCrO4 spinel (Table IV).
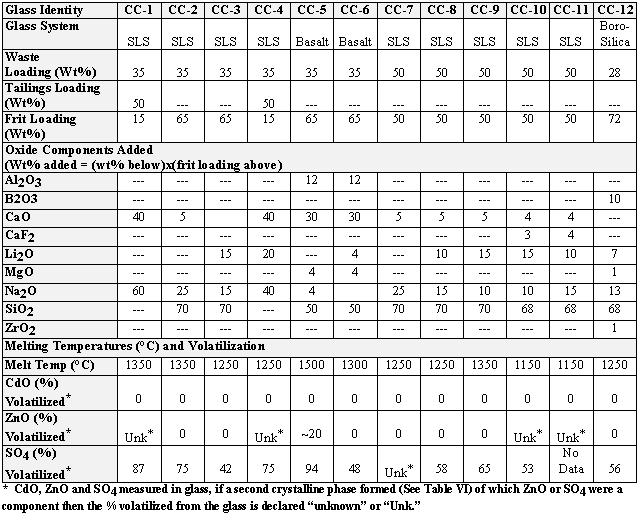
The remaining seven SLS glasses were made with combinations of high surface area silica (SiO2) known to increase the solubility of Zn and other heavy metals in the glass (C.M. Jantzen, J.B. Pickett, and H.L. Martin, "Method for Treating Materials for Solidification", Reactive Additive Stabilization Process (RASP™), U.S. Patent 5,434,333 (July 18, 1995)), and with calcium carbonate (CaCO3), sodium carbonate (Na2CO3), lithium carbonate (Li2CO3), and calcium fluoride (CaF2) as fluxing agents (Table III). The glasses made at 35% waste loading, CC-2 and CC-3, were melted at 1350°C and 1250°C respectively (Table III). Glass CC-2 contained only one alkali, Na2O, while CC-3 had both Na2O and Li2O present as fluxing agents (Table III). Both CC-2 and CC-3 SLS formulations made excellent homogeneous glass (Table IV).
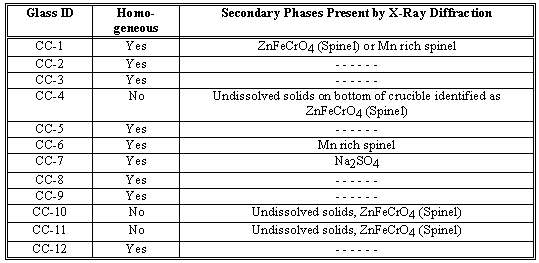
Glasses CC-7, CC-8, and CC-9 contained 50 wt% waste and melted at 1250°C, 1250°C, and 1350°C respectively. Glass CC-7 had only Na2O present as a fluxing agent. Glass CC-7 was not homogeneous as a secondary sulfate salt phase, Na2SO4, formed which floated on the glass surface. Both CC-8 and CC-9 had Na2O and Li2O present as fluxing agents and both produced excellent quality glass.
When CaF2 was partially substituted for the CaO added to glass formulations CC-8 and CC-9 as a third fluxing agent, the glasses produced (CC-10 and CC-11) were not homogeneous (Table IV). Glasses CC-10 and CC-11 contained undissolved waste solids, primarily ZnFeCrO4 spinel. The homogeneous SLS glass formulations are CC-1,2,3 and CC-8,9. These glasses may be melted in any type of Joule or gas-fired melter. Glass CC-7 produced a molten Na2SO4 salt phase on the glass surface. Glass CC-7 could be vitrified in certain types of melters (such as the TVS) equipped to "tap-off" molten salt layers which form on melt pool surfaces and avoid safety hazards associated with salt layer formation.
Two of the twelve glasses (CC-5 and CC-6) formulated were in the basalt glass forming system (Table III). Glass CC-5 was melted at 1500°C while glass CC-6 was melted at 1300°C (Table III). Glass CC-5 was an excellent homogeneous glass (Table IV). Glass C-6 had a very slight film on the glass surface and some solids remained in the crucible. The undissolved solids and film were was identified as a manganese rich spinel (Table IV) which is not a component of the original waste.
Only one borosilicate glass was formulated with the mining sludge. The high iron and high manganese content of the sludge is similar to the high iron and manganese levels in SRS high level waste: the borosilicate glass Frit 165 was used as a glass forming additive. A waste loading of 28 wt% (Table III) and a frit loading of 72 wt% was used. The composition of the Frit 165 is given in Table III. The mining sludge made an excellent high iron containing borosilicate glass. which can be melted in most conventional melters at 1150°C.
Volatilization
Chemical analyses of the glass product indicated that both the CdO and the ZnO in the waste sludge were retained in the glass (Table III) regardless of the glass processing temperature. Sulfate is a component of the mining sludge (4.8 wt%) and the mill tailings (3.29 wt%). The SO4 content of the sludge/mill tailings was partially retained in the glass and partially vaporized as SO2 or SO3 gas. Volatilization was almost a linear function of melt temperature (Figure 1). Volatilization of some SO4 occurred from every glass melted regardless of the melt temperature. Since the mill tailings contain a significant amount of SO4 as the mineral gypsum, the use of the mill tailings would create a significantly larger SO2/SO3 gaseous emission in the off-gas system during vitrification. Therefore, use of high sulfate containing (gypsum containing) mill tailings was not recommended.
Volume Reductions
Large volume reductions (87-93%) were realized upon vitrification of the mining waste sludge due to the vaporization of water (~86.7 wt%), densification from a sludge of ~1 g/cc to a glass of ~2.7 g/cc, and volatilization of OH-, NOx, CO2 , SO4 from hydroxide, nitrate, carbonate, and sulfate species present in the sludge. The volume reductions for a nominal waste loadings of 35 wt% sludge on a dry oxide basis gives a 90% volume reduction. Therefore, 1000 kgs of sludge produces 281 kgs of waste glass. When compared to alternative stabilization technologies such as cement which give a volume increase of 150-200% the volume reductions given by vitrification are even larger, e.g. for this case study 1,440,000 "55 gallon drums" of cement would be produced compared to 7930 "55 gallon drums" of glass on a yearly basis. That is, 18 drums of waste cement would be produced for every drum of waste glass. In addition, the vitrified waste can be recycled whereas the cement waste form cannot.
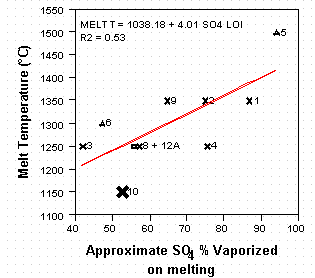
Figure 1. Relationship of SO4 vaporization
to melt temperature where LOI is
the Loss-on-Ignition at the melt temperature indicated.
Recycle Markets
The use of treated materials containing hazardous metals has been previously considered by the EPA for residues remaining from High Temperature Metal Recovery (HTMR) operations (Standards for the Management and Use of Slag Residues Derived From HTMR Treatment of K061,K062, and F006 Wastes. 59 FR 67256, December 29, 1994). In this proposal dealing with the use of residues from HTMR, the EPA proposed that such wastes would not be subject to hazardous waste disposal regulations - if they meet 3 conditions:
These treated materials could be used for recycling as (1) covered sub-base materials (e.g., in construction of paved roads, parking lots, and driveways), (2) additive ingredients in cement or concrete /asphalt mixtures, (3) top grade or surfacing materials, e.g., in construction of roads, parking lots, and driveways (glassphalt or glasscrete) (Uses for glassphalt (crushed glass is added to asphalt to improve its rigidity) can be found on the WWW and include air-port runways, highways in Virginia, New York City, and Los Angeles most notable of which is Hollywood Blvd (glassphalt gives it sparkle)), and as anti-skid / de-icing materials. Note; the current regulations for the HTMR residues specify that the wastes are NOT hazardous wastes, if they meet the exclusion requirements - but they MUST be disposed to Sub-Title D landfills (i.e.; at this time the proposed reuse (59 FR 67256, 12/29/94) for the HTMR residues has not been adopted). Therefore, if the vitrified treated mining wastes meet the two restrictions above (inseparable and LDR standards) then the mining waste glass should be allowed into the recycle market.
Conclusions
Mining wastes can be vitrified into a glass that meets the EPA regulations for High Temperature Metal Recovery (HTMR) operations. This allows the vitrified product to enter the recycle market.
Acknowledgement
This paper was prepared in connection with work done under Contract No. DE-AC09-96SR18500 with the U.S. Department of Energy.
References